Q: 4. For which compound is cis/trans isomerism possible? a. Ethene b. 2-butene C. 2-methyl-2-butene d.…
A:
Q: Decide whether these proposed Lewis structures are reasonable. proposed Lewis structure |:N—N= : 0:…
A:
Q: Use the drop-down selection boxes to rank the IR absorptions of the coloured bonds in terms of…
A: IR spectroscopy is mainly used for identification of functional group present in the molecules. Each…
Q: mCPBA Select to View feedback Q Please select a drawing or reagent from the question area Incorrect,…
A: Epoxidation is the oxidation reaction of alkene Or alkynes in which one oxygen atom is inserted…
Q: 16. Which of the following compounds is most polar? a. CH3CH₂CH₂OH b. CH3CH₂C(O)OH C. CH3C(O)OCH₂CH3…
A: Polarity of the organic compound is depends upon the strength of compound making hydrogen bond.
Q: Draw and discuss in details the Pourbaix plot representing the following reactions with a…
A: A Pourbaix diagram is a plot of the equilibrium potential of electrochemical reactions against pH.It…
Q: Be sure to answer all parts. Find the pH of the two equivalence points and the volume (mL) of 0.0784…
A: Titration of 17.3 mL of 0.130 M H2CO3 with 0.0784 M KOH.We have to find out the pH of the two…
Q: Explain using an example the relationship of chemical potential to phase equilibrium.
A: Chemical potential is a thermodynamic property that describes the potential energy of a substance to…
Q: Predict the products (if any) of the following reactions and write the molecular, complete ionic and…
A: Aqueous copper perchlorate and potassium bromide reacts to form copper bromide and potassium…
Q: A 25.0 mL solution of HNO3 is neutralized with 15.7 mL of 0.250 M Ba(OH)2. What is the concentration…
A: In the given question, we have to find the concentration of HNO3 solution.
Q: ) You measure the pH of your solution to be 2.1. What is the [H3O+]? State your answer in scientific…
A: As this is multiparts question, as per rule I am going to solve only first question. If you need…
Q: [Review Topics] [References] Use the References to access important values if needed for this…
A: Osmolarity refers to the number of solute particles per 1 L of solvent
Q: What is the reactant that is classified as Bronsted-Lowry acid? CO3²¯(aq) + H₂O(l) ↔ HCO3¯(aq) +…
A: Bronsted Lowry acid is a substance which donates a proton to other compound and form conjugate base.…
Q: give the common name of this compound
A: The common name is a non-systematic name given to a chemical compound that is widely used in…
Q: ct the most stable alkene 2,3-dimethyl-2-pentene -pentene -2-methyl-2-pentene { * 1-pentene
A: The stability of the given alkene can be decided based on the number of substituents attached…
Q: Question 5 of 32 Determine the value of Ksp for Mg(CN), by constructing an ICE table, writing the…
A: Given,The molar solubility of MgCN2 is 1.4 × 10-5 MRequired, The value of Ksp for…
Q: What is the IUBAC name CH3 I CH3----CH----CH2CH3
A: Answer: Given compound is an alkane and we have to write down its name following the IUPAC…
Q: What is the IUPAC name CH3 CH3 I I CH2=CH-CH-CH-CH3
A: The given molecule is an alkene with several methyl substituents.
Q: X-Y Br The transformation above can be performed with some reagent or combination of the reagents…
A: We have to identify sequence of reaction
Q: true or false? - 3 The enthalpy changes for the reaction CO₂(g) + 3H₂(g) CH₂OH(g) + H₂O(g) exceeds…
A: The enthalpy change of a reaction is given by the difference between sum of enthalpies of products…
Q: Write reasons for the regioselectivity of hydroboration-oxidation of alkene by drawing the reaction…
A: Hydroboration is the addition of borane to alkene, alkyne to form the alkyl borane, alkenyl borane.…
Q: If K₁ for NX3 is 3.5x10-6, what is the pOH of a 0.175 M aqueous solution of NX3? Express your answer…
A: In the question, it is given that Kb of NX3 = 3.5×10-6 We need to find pOH of 0.175M aqueous NX3.
Q: A 60 g Butterfinger candy bar has 270 Cal, which is ____ kJ. a) 1.88 x 105 kJ b) 1130 kJ c) 1.07 x…
A: Given : Calories in the candy bar = 270 Cal
Q: 0:08 AM Thu Mar 23 Problem 33 of 23 Br2 (1 equiv) Draw the product of the reaction shown below.…
A:
Q: If a sample transmits 74.8% of the light you shine on it, what is the absorbance of the sample?
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: QUESTION 7 One mole of particles of any substance contains how many particles? 3 x 10-10 particles 3…
A: 7) The value of the mole is equal to the number of atoms in exactly 12 grams of pure carbon-12.
Q: a) Complete and balance Ba(CN)2(aq) + b) What type of reaction is this? c) Is this a redox reaction,…
A:
Q: provide the IUPAC name of the product formed when 2-butanol is dehydrated
A: When 2 - butanol is dehydrated means it’s water molecule is removed out than butene is formed…
Q: Consider the weak acid, HA, in water as shown in the reaction below. The [HA] initially is 0.150 M…
A: Ka is acid dissociation constant. For weak acids we can calculate the pH of a weak acid.
Q: Consider the weak base, B -, in water as shown in the reaction below. The [B -] initially is 0.340 M…
A:
Q: 3) What volume (in milliliters) of 0.1M AgNO3 would be needed to completely react with 2.50 g zinc?…
A:
Q: 5. A buffer is made with 0.147 moles of acetic acid and 0.185 moles of sodium acetate to make 2.00…
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base . it…
Q: - ] 3 Consider the reaction 1205(g) + 5 CO(g) → 5CO2(g) + 2(g) 80.0 grams of iodine pentoxide, 1205…
A: Number of moles is calculated by dividing the given mass with molecular mass. In the given question…
Q: How many grams of ammonia vapor gas (NH3) are in a 500.0 mL sealed flask at 68.5°C and 5.04 atm?
A: The ideal gas equation for the relation between pressure (P), volume (V) and temperature (T) is:…
Q: 7. What is the IUPAC name of the following compound? ic 8. What is the IUPAC name of the following…
A: We have to determine the IUPAC name of the given structures
Q: Draw the skeletal ("line") structure of an isomer of this molecule:
A: Isomers are those compounds having same molecular formula but different structures or the different…
Q: FH₂C FH₂C CH3OH dil. H₂SO4 ? 1. Hg(OAc)2, CH3OH 2. NaBH4 ?
A:
Q: compute for the heat gained, specific heat of metal material: aluminum mass of metal: 8.8399g…
A: Given : Mass of metal = 8.8399g Initial temp of metal : 100 °C Mass of water = 18.4120 g Initial…
Q: What best describes a solution with a [OH -] of 2.068x 10 -12 M? Acidic Basic Neutral
A:
Q: In the reaction 2AI +3F22AIF3, what would be the actual yield if 50.0g of fluorine reacts with 50.0g…
A: Reaction is 2 Al + 3 F2→2 AlF3 2 mole Al react with 3 mole F2 to form 2 mole AlF3.
Q: The NO molecule has an unpaired electron in it π* orbital which will be donate to an electron-rich…
A: We have to tell about the N-O bond length, when NO is coordinate to an electron rich transition…
Q: 6. Alkenes cannot undergo which type of reaction? a. Addition b. Polymerization C. Oxidation d.…
A: Alkenes are unsaturated hydrocarbons that contain a carbon-carbon double bond. The double bond makes…
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A: It is based on the different types of the reaction Here we are required to balanced molecular…
Q: 1.35mol of copper reacts with an excess of silver nitrate according to the reaction above. from this…
A:
Q: 4. A 0.522 g sample of an unknown metal (X) reacted with hydrochloric acid according to the balanced…
A: Given : Mass of metal(m) = 0.522 g Volume of H2 gas(V) = 232 ml = 0.232 L Pressure(P) = 775 mm Hg…
Q: 10. Phosphoric acid is used in rust-remover solutions. The aqueous acid is available for purchase by…
A:
Q: Teage reaction. OH ✓
A: In the given reaction -OH is not a good leaving group. Hence it should be converted into a good…
Q: 5. The reaction, 2A + 2B C + D, has a rate constant of 6.0x10-3 L² mol-2 s¹ at 10°C. If its…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A:
Q: Propose an efficient synthesis for the following transformation: Br The transformation above can be…
A: The question is based on the concept of organic synthesis . we need to synthesize the product from…
D.7.
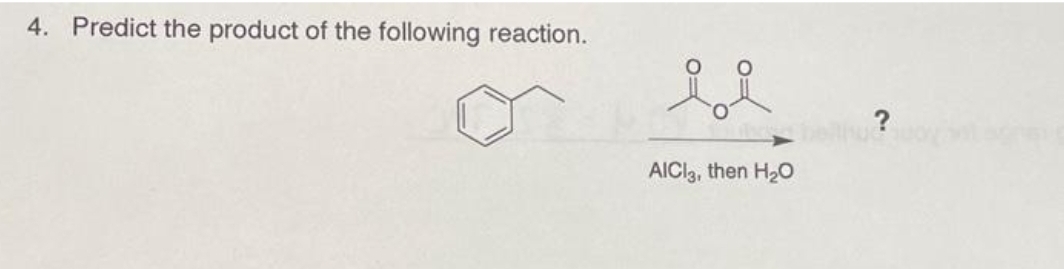
Step by step
Solved in 2 steps with 2 images


