5- To know the number of neighboring water molecules linked by H-bond to a molecule, and to describe the structural organization of the ice, it is necessary to add neighboring meshes. From figure 4.2, how many neighboring molecules does a water molecule have, how are they linked ? What do the hands and feet in Figure 4.2a represent to you? a)b Liaison hydrogène dans l'eau J.C. Bünzli Lausanne O Philip Ball Model representing hydrogen bonds (© Philip Ball) Figure 4.2: schematic representation of the local environment of a water molecule.
5- To know the number of neighboring water molecules linked by H-bond to a molecule, and to describe the structural organization of the ice, it is necessary to add neighboring meshes. From figure 4.2, how many neighboring molecules does a water molecule have, how are they linked ? What do the hands and feet in Figure 4.2a represent to you? a)b Liaison hydrogène dans l'eau J.C. Bünzli Lausanne O Philip Ball Model representing hydrogen bonds (© Philip Ball) Figure 4.2: schematic representation of the local environment of a water molecule.
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter8: Molecules And Materials
Section: Chapter Questions
Problem 8.88PAE
Related questions
Question
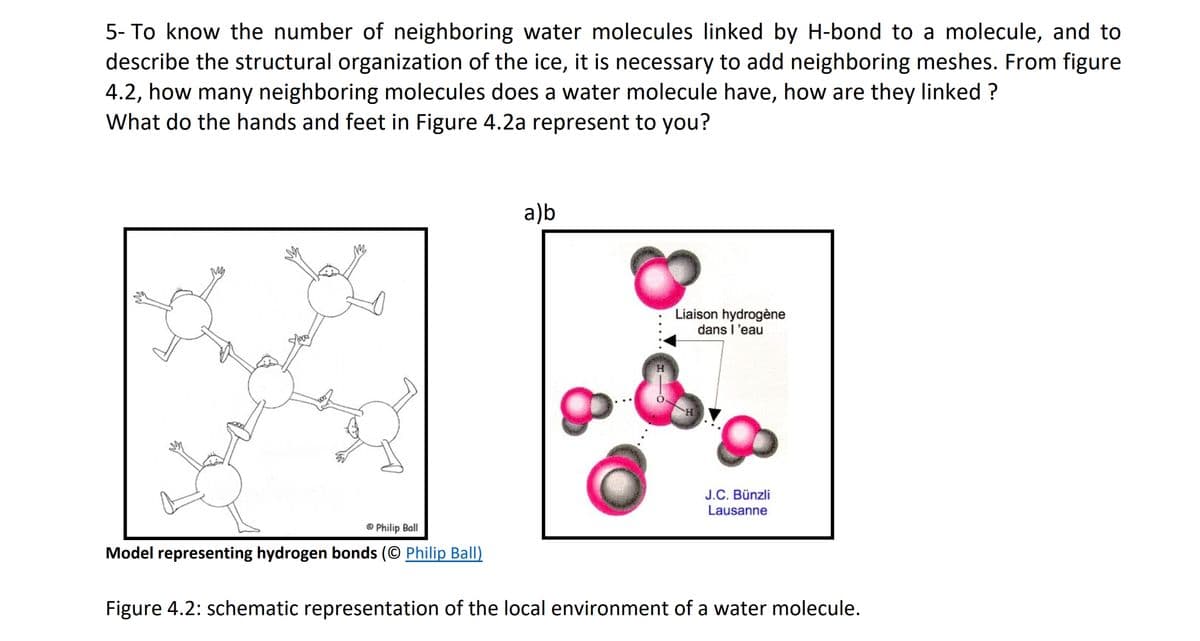
Transcribed Image Text:5- To know the number of neighboring water molecules linked by H-bond to a molecule, and to
describe the structural organization of the ice, it is necessary to add neighboring meshes. From figure
4.2, how many neighboring molecules does a water molecule have, how are they linked ?
What do the hands and feet in Figure 4.2a represent to you?
a)b
Liaison hydrogène
dans l'eau
H.
J.C. Bünzli
Lausanne
© Philip Ball
Model representing hydrogen bonds (© Philip Ball)
Figure 4.2: schematic representation of the local environment of a water molecule.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax