559048ad75799f5abe16e1aff#10001 MReview | Constants Periodic Table On the basis of periodic trends, choose the larger atom in each pair (if possible): Match the items in the left column to the appropriate blanks in the sentences on the right. Make certain each sentence is complete before submitting your answer. Re F Of Ge or Po, the larger atom is Se Of Cr or W, the larger atom is Po Of F or Se, the larger atom is Sn Ge Of Sn or I, the larger atom is not predictable I. W Cr Pearson All rights reserved. | Terms of Use | Privacy Policy I Permissions | Contact Us I 8:06 PM 80°F Raining now 10/7/2021 8.
559048ad75799f5abe16e1aff#10001 MReview | Constants Periodic Table On the basis of periodic trends, choose the larger atom in each pair (if possible): Match the items in the left column to the appropriate blanks in the sentences on the right. Make certain each sentence is complete before submitting your answer. Re F Of Ge or Po, the larger atom is Se Of Cr or W, the larger atom is Po Of F or Se, the larger atom is Sn Ge Of Sn or I, the larger atom is not predictable I. W Cr Pearson All rights reserved. | Terms of Use | Privacy Policy I Permissions | Contact Us I 8:06 PM 80°F Raining now 10/7/2021 8.
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter6: Electronic Structure And The Periodic Table
Section: Chapter Questions
Problem 67QAP: Write the symbol of each element described below. (a) largest atomic radius in Group 1 (b) smallest...
Related questions
Question
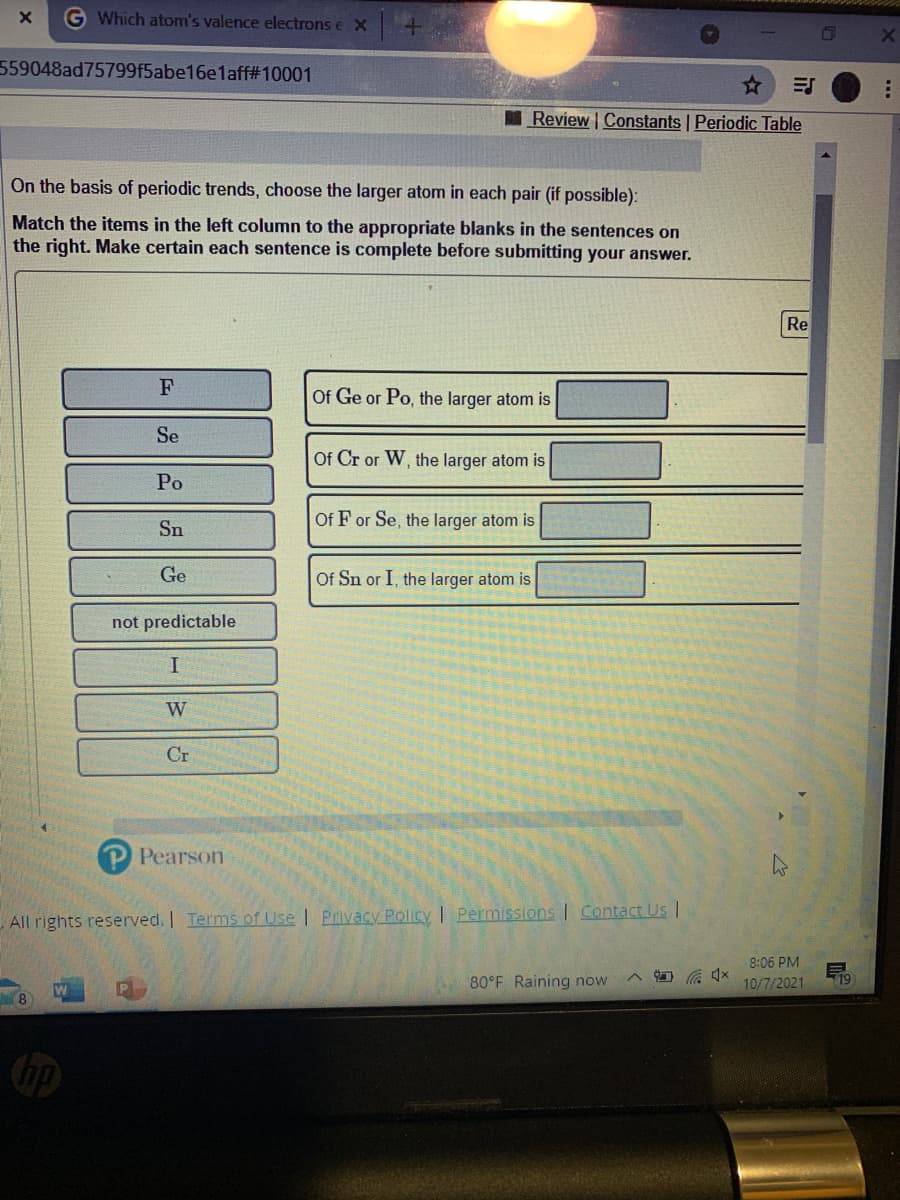
Transcribed Image Text:G Which atom's valence electrons e X
559048ad75799f5abe16e1aff#10001
Review Constants | Periodic Table
On the basis of periodic trends, choose the larger atom in each pair (if possible):
Match the items in the left column to the appropriate blanks in the sentences on
the right. Make certain each sentence is complete before submitting your answer.
Re
F
Of Ge or Po, the larger atom is
Se
Of Cr or W, the larger atom is
Po
Of F or Se, the larger atom is
Sn
Ge
Of Sn or I, the larger atom is
not predictable
W
Cr
Pearson
All rights reserved. I Terms of Use | Privacy Policy I Permissions | Contact Us |
8:06 PM
80°F Raining now
10/7/2021
19
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning