Q: Energy cannot be created nor destroyed, but it can be transferred between a system and its…
A: Internal energy :Internal energy is state property and extensive property and it is sum of total…
Q: The picture below represents the overlap of two px orbitals to form a pi star (π*) antibond. The…
A: And antibonding Molecular Orbital is formed by the overlap of the lobes of opposite phases of the…
Q: Choose the aqueous solution with the lowest vapor pressure. These are all solutions of nonvolatile…
A: Lowering of vapor pressure is a colligative property. Colligative properties are those properties…
Q: Five students performed a Kjeldahl nitrogen analysis of a protein sample. The weight % nitrogen…
A: The weight percentage values of nitrogen determined by the Kjeldahl method are given as 15.5, 14.0,…
Q: How many grams of crystal violet are needed to prepare 10.00 mL of 6.824 * 10-6 M solution of…
A: Solute: The minor component in the solution.Solvent: The major component in the solution.Molarity is…
Q: 9. Draw the sigma bond that forms from an S orbital bonding with an sp orbital.
A: One of the lobes of the sp2 hybridised orbital undergoes head-on overlap with s-orbital to form a…
Q: PC15(g) PC13(g) + Cl₂(g) has Kp = 1.84 at a given temperature. What is the value of Kp for the…
A:
Q: The image below shows a portion of a 'H NMR spectrum (ppm). Choose the partial structure that best…
A: The given portion of the 1H-NMR spectrum has one doublet near 0.9 ppm and one septet near 3.9…
Q: The atomic number of the element neon (Ne) is 10. How many electrons does a neutral atom of neon…
A: Given, The atomic number of the element neon ( Ne ) is 10
Q: Q.3. Write Lewis structures for the following compounds. Show formal charges, if any. a) C₂H₂O b)…
A: A Lewis dot structure is defined as a set of atoms surrounded by electrons in which the electrons…
Q: 1. Write the IUPAC names for the following two structures:
A: IUPAC nomenclature used for the systematic naming of the organic molecules.According to IUPAC…
Q: When the buret in the example delivers from 0.06 mL to 40.00 mL. What is the real volume delivered?…
A: Check the volume of the solution delivered from the buret from the given mark of the buret.
Q: How many molecules of carbon monoxide (CO) will be formed when charcoal reacts with 1.505*10^(23)…
A: Balanced chemical equation is2C(s) + O2 (g) ----> 2CO(g)
Q: draw the structural formula for the following molecule written in condensed form: CHCCH(CH3)CH2CH3
A: A structural formula of organic compound is the representation of the compound where each atom is…
Q: A chemist working as a safety inspector finds an unmarked bottle in a lab cabinet. A note on the…
A: Given Volume of unknown liquid = V =1049 cm31 cm3 = 1mL1049 cm3 = 1049 mL = VMass of unknown liquid…
Q: Solve: 0.025 x 4.62 3.44 0.340 0.00335 0.034 0.033575581
A: We have been asked two qyestions related to significant figures In one question, we have been given…
Q: Which of the listed presentations are resonance structures for the cation in the image: Choose...…
A: Resonance structures are representations of different Lewis structures of same molecule.By moving…
Q: Select the common name for each of the following two dicarboxylic acids: HOOCCOOH adipic acid oxalic…
A: The given dicarboxylic acids are,
Q: The heat capacity of an object indicates how much energy that object can absorb for a given increase…
A:
Q: 13.26 Assign an IUPAC name for each of the following compounds: X a. b. d. ● شاه 8. SH e, o f. S…
A: " Since you have posted a question with multiple subparts, we will provide the solution only to the…
Q: Collected Volume sodium carbonate (mL) Molarity sodium carbonate (M) Volume calcium chloride (mL)…
A: The reaction taking place between Sodium carbonate () and Calcium chloride () is -This results in…
Q: The maximum amount of chromium(III) hydroxide that will dissolve in a 0.260 M chromium(III) nitrate…
A: The capability of a solute or a substance to interact with solvent and form a solution is called…
Q: Using factor label calculations, convert 5.00 g/100 mL glucose to lbs / gallon. (2.205 lb = 1 kg, 1…
A: glucoseWe have to convert into .
Q: 6. Use Hess's Law and the following data: Sro(s) à Sr (s) + 1/20₂(g) SrO(s)+ CO₂(g) à SrCO3(s) 2…
A:
Q: For each pair of compounds listed, check the box next to the one with the higher boiling point. high
A: Boiling point is a temperature where a liquid starts to boil or the vapour pressure of liquid…
Q: A 13C NMR spectrum is shown for a molecule with the molecular formula of C4H8O2. Draw the structure…
A:
Q: A new drug has been discovered and must be formulated as an IV injection. The drug is a non-…
A: Concertation in (w/v)% :- This percentage weight by volume formula is expressed as %w/v, meaning…
Q: What is the correct answer to the following expression? x 5.45 10-10 +5.74 x 10-12 Selected Answer:…
A: Given ,Expression : Objective : To find the sum of given expression
Q: A 759.2 ng sample of an unknown radioactive substance was placed in storage and its mass measured…
A:
Q: Draw an orbital diagram for the element nickel.
A: Atomic number of Nickel is 28andElectron configuration of Ni: 1s2 2s2 2p6 3s2 3p6 4s2 3d8
Q: A calibration curve was created to determine the quantity of protein in a solution. The calibration…
A: Corrected absorbance of any solution is found by deducting the absorbance of the blank solution from…
Q: Use both the van der Waals and the Redlich-Kwong equations to calculate the molar volume of CO at…
A: Temperature = 197 KPressure = 1026 barVan der Waals equation, a = 1.4734 dm3.bar.mol-2 Van der Waals…
Q: Draw all stereoisomers that are possible for the following unsaturated alcohol. CH3 CH₂…
A: Stereoisomers are molecules with identical molecular formulas but differ in the position or…
Q: which of the following has electrons in the 5 p shell? Ba Cd As Cr. са
A: Electronic configuration can be define as the arrangement of electrons systematically into various…
Q: Draw the two major products obtained in the reaction shown. + Br₂ (one equivalent) CH₂Cl₂
A: Given reaction is bromination reaction. Bromination reaction is electrophilic addition reaction of…
Q: Give clear detailed Solution with explanation needed..don't give Handwritten answer
A: We know that alkyl compounds react with bromine in presence of light to form the respective alkyl…
Q: Write SMILES formula for an anion presented with the molecular formula [CN]-.
A: Given Molecular formula: [CN]-To determine: SMILES formula for the above mentioned anion
Q: Rank the following conformers of 1-propanol in DECREASING order of stability by inserting a number…
A: The first conformer is gauche presentation,Second is fully eclipsed form,third is anti formand last…
Q: 4) The molality of naphthalene in a solution of naphthalene (C₁oHs, MW= 128.18 g/mol) and…
A: The colligative properties are those properties of solutions that depend on the number of solute…
Q: Draw bond-line formula for the listed compounds showing lone pairs of electrons and formal charge on…
A: It is a representation of molecular structure in which covalent bonds are represented by one line…
Q: What is the value of Kc for this reaction at the same temperature?
A: Kc = [Products] / [Reactants]
Q: When 12.7 and 97.0 are multiplied, the answer should be reported to significant digit(s). The…
A: Rules for determining the number of significant figures in an answer :The result of an addition or…
Q: (K) 1-bromobutane кон, н,о (A) Na (B) CH, CH₂Br (C) Mg, ether (1)CH,…C…CH,CH, (2) H₂O+ (J) (D)…
A:
Q: How many stereoisomers are possible for 2,4-heptadiene? CH₂-CH=CH-CH=CH-CH₂-CH₂
A:
Q: IS tell measured in the hectare is equal to 10,00 often measured in acres conversions and those g…
A:
Q: Complete the reaction below CH₂ + Cl₂ CH₂Cl₂
A: The addition of chlorine to an alkene in a nonpolar solvent produces vicinal dichloro alkane.
Q: An aqueous solution of isopropanol (MM = 60.10 g/mol) has a molality of 6.05 m and a density of…
A:
Q: Set up the problem and calculate the mass of 16.9 mL of copper. See the table below for the density…
A: We have, volume of copper = 16.9 mLmass of copper =? gramsDensity of copper = 8.94 g/mLDensity is…
Q: For the gas phase isomerization of cis-cyanostyrene, cis-C, H, CH=CHCN trans-C, H, CH=CHCN -1 the…
A: The rate constant of a reaction is temperature dependent.It varies as the temperature…
Q: Do any of the atoms in the above compounds have a formal charge?
A: The octet rule states that the elements of the s and p blocks try to attain the nearest noble gas…
6) propose a mechanism with arrows for the following transformations.
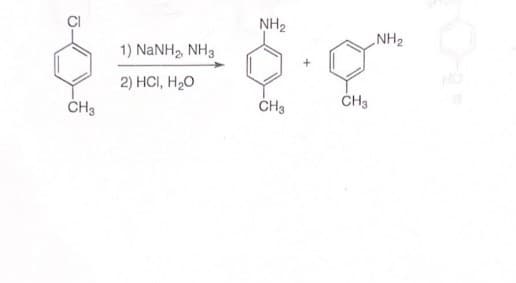
Step by step
Solved in 3 steps with 1 images


