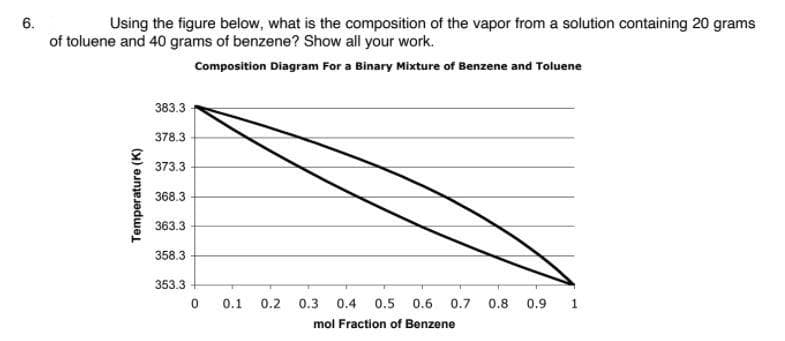
6. Using the figure below, what is the composition of the vapor from a solution containing 20 grams of toluene and 40 grams of benzene? Show all your work. Composition Diagram For a Binary Mixture of Benzene and Toluene 383.3 378.3 373.3 368.3 363.3 358.3 353.3 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1. mol Fraction of Benzene Temperature (K)
Q: Q1: Indicate whether the following statements are true or false ,correct the false and give a proper…
A:
Q: 3. When solutions containing 1.0000 g of BaCl2 and 1.0000 g of NazSO4 were mixed, solid BaSO4…
A: Note - Since you have posted a question with multiple sub-parts, we are supposed to solve only the…
Q: It is important to check the information attached to a product before using it.* True False Consists…
A:
Q: A chemist mixes 94.1 g of acetone with 58.0 g of thiophene and 82.4 g of methanol. Calculate the…
A: Interpretation- To determine the percentage by mass for each component , component are given in the…
Q: The volume of a solution was measured in a graduated cylinder (shown below), and the density of…
A: We know that density = mass / volume The volume of solution is measured in graduated cylinder The…
Q: A calibration curve of ZnCl2 conductivity (y-axis) vs concentration (mass percent, x-axis) is made.…
A: In the linear regression plot, the X-axis represents the mass percent of ZnCl2. The Y-axis…
Q: Newly produced beer must be checked to see if it meets predetermined standards. The percentage of…
A: We need to calculate the average percentage by volume of ethanol for the batch: The batch contains 4…
Q: Solve for empty boxes.
A: The values that should appear in the empty boxes of the given table are needed to be calculated.
Q: Part 1 – Preparation of CaCO3(s) The following is a procedure that was theoretically performed…
A: Given: Volume of CaCl2 = 10 mL concentration of CaCl2 = 1.0 M Volume of K2CO3 = 25 mL concentration…
Q: What does simple distillation data from the same mixture would look like? 2. Why does the…
A: As you not specify so I am giving answer of second question collecting the cyclohexene fraction…
Q: After performing precipitation gravimetry, Asta placed the weighing form inside the crucible, heated…
A:
Q: An aqueous solution of potassium sulfate, K2SO, is made by dissolving 27.6 grams of potassium…
A: W/V% =( weight of solute/ volume of solution)*100
Q: A 100 mg vitamin C tablet weighed 1.681 g. A student crushed the tablet and transferred 0.150 g of…
A: Given the weight of 100 mg Vitamic C (ascorbic acid) tablet = 1.681 g 0.150 g of the above tablet…
Q: Using the attached curve, If 65 g of NH4Cl are added to 100 g of water at 50 °C, how many grams of…
A: All known compounds can be dissolve in certain solvents whereas they remain insoluble in other…
Q: When 0.501 g of sucrose (table sugar, white microcrystalline solid) is dissolved in water (clear…
A: Quantitative data are data which are obtained experimentally and helps In quantification of element…
Q: ayer Mass of Erlenmeyer Flask Calcium Hydroxide olution (lime water) Mass of Calcium Hydroxide…
A: Titration of Calcium Hydroxide (Ca(OH)2) with Hydrochloric acid (HCl) is- Ca(OH)2 + 2HCl…
Q: Low of constant composition
A: In this question, we will see the defination of Law of constant composition and it's example. You…
Q: After performing precipitation gravimetry, Asta placed the weighing form inside the crucible, heated…
A: The average of these masses is : 23.1136 + 23.2125 + 23.4120 + 23.11394 = 23.2130
Q: Complete the following problems. Show the correct number of significant figures and units in your…
A:
Q: 16. Water and acetonitrile, CH3CN, are miscible (can be mixed in any proportions). However, when…
A: Ans Water and methylcyanide are miscible with each other. Total volume of the solution = Volume of…
Q: Mass of Erlenmeyer Flask (g) 24.33 24.37 24.44 Mass of Erlenmeyer Flask + Calcium Hydroxide…
A: The comment should be: We’ll answer the first question since the exact one wasn’t specified. Please…
Q: Allison dissolves as much as possible of a certain solid in a quantity of water at room temperature;…
A: Solubility is the mass of the solute dissolved in particular amount of solution .
Q: A sample is said to be homogeneous if 1. OThe sample has a different composition in different parts…
A: Correct answer is (2.) The sample is completely mixed up. Homogenous mixture has uniform mixture and…
Q: 3. When solutions containing 1.0000 g of BaCl2 and 1.0000 g of NazSO4 were mixed, solid BaSO4…
A: Given that : The mass of BaCl2 used = 1.000 g The mass of Na2SO4 used = 1.000 g The molar mass of…
Q: 5. Determining the percent sulfuric acid in an unknown solution. Experimental Procedure and Results:…
A: Given: Mass of empty 50 mL beaker = 52.5 g Mass of beaker + unknown solution = 66.55 g Drops of NaOH…
Q: A nitric acid solution is labelled 6.0% W/W nitric acid by mass. What mass, in kg, of pure solute is…
A: Given, Mass of solution = 1.84 Kg Concentration % (w/w) = 6.0 % Required, Mass of pure solute = ?
Q: Help me solve the following problem correctly and completely. Express all answers to the nearest…
A: H2SO4 is a diprotic acid.The molar mass of H2SO4 is 98.078 g/mol.It provides 2 moles of H+ ions for…
Q: A heterogeneous mixture Is the one which O has one phase O has a uniform composition throughout O…
A: The question is based on the concept of solutions . We have to identify which of the given…
Q: best value for percent purity and how did you arrive at the best value
A: The best value is calculated from the average of the good data values.
Q: You record the following data during the Qualitative and Quantitative Characterization of Aspirin…
A: A numerical problem based on concentration terms, which is to be accomplished.
Q: A. Standardization of NaOH Solution: Sample A Sample B Sample C 0.100m 0.100m 0.100m 1. Molarity of…
A: Given: The volume of 0.100 M HCl used for the determination of molarity of NaOH is 25.00 mL.
Q: The Results for SVI Calculation of a Mixed Liquor Sample Weight of Filter Paper = 0.603 g Weight of…
A:
Q: You have a solution of NaCl that has a molarity of 3.359 M. You want to make a dilute solution of…
A:
Q: A student dissolves a Jefferson nickel to make 100.00 mL of solution in a volumetric flask. The…
A: Mass of Copper in Jefferson nickel used for making the first solution is taken as ‘x’ g.Hence,…
Q: Instructions: solve as neatly as possible and show complete solution. all values must include…
A:
Q: Data for 5.0% NaCI Solution Mass of 50mL beaker 28.93 g Mass of beaker + 10.00mL of 5.0% NaCl…
A: From given Density of 5% NaCl and 10% NaCl solutions are determined by finding its mass and…
Q: Be sure to answer all parts. Fluoridation is the process of adding fluorine compounds to drinking…
A: One person consumes 140gallon 1 gallon is equal to 3.785L The density of water = 1g/ml which will…
Q: A chemist mixes 59.3 g of acetone with 97.8 g of 2-ethyltoluene and 47.1 g of isopropenylbenzene.…
A: Mass percent is defined as the mass of the substance divided by the total mass of the mixture ,…
Q: A homogeneous mixture has non-uniformly distributed particles a uniform composition throughout O not…
A: There are generally two types of matter which are considered as pure substance and mixture. Pure…
Q: using exactly 5.00 mL of 0.0400 M stock CuSO4 solution. Add 100 mL of water. Data for Part I Mass…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: I need help finding the %Composition for %cychohexane and %toluene. The formula (area of peak/total…
A: To determine: % composition for Cyclohexane and toluene
Q: 5. A 100-kilogram salt solution originally 4% by weight. Salt in water is boiled to reduce water…
A:
Q: Consider the table below: Compound Density Ether 0.8415 g/mL Dichloromethane 1.311 g/mL Water 1.00…
A: From the given data of densities of ether, dichloromethane and water – it is clear that density of…
Q: Sample 1 Sample 2 Sample 3 Mass of Erlenmeyer Flask (g) 24.33 24.37 24.44 Mass of Erlenmeyer…
A: Ca(OH)2 ----> Ca2+ + 2 -OH 2 -OH + 2 HCl -----> 2H2O + 2Cl- net reaction Ca(OH)2 + 2 HCl…
Q: The density of a series of solutions with different concentrations of NaCl is measured. The results…
A: Density is the measure of the mass of the matter occupied per unit volume of the container. Here,…
Q: The density of a series of solutions with different concentrations of NaCl is measured. The results…
A: Plot of density (g/mL) Vs concentration (NaCl % by weight) is given by,
Q: What is the average percentage by volume of ethanol for this batch?
A:
Q: A physician requests that an iso-alcoholic elixir containing 30.0% alcohol be prepared. How many mL…
A: Given, Total volume of solution= 1000ml % iso-alcohol elixir for final solution = 30% % low…
Q: 1. An investigator is interested in identifying which metals are present in seaweed found off the…
A: The detection and analysis of metal ions in a specific source can be best accomplished by employing…
Q: Why is the zinc powder weighed roughly while CuSO4 solution is pipetted?
A: A hydrate is a substance that contains water or its constituent elements. Hydrate are any compound…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- 1a. Use the given table to construct a temperature-composition diagram for cyclohexane Toluene mixture. Based on the plot, what is the composition of the distillate obtained by Simple distillation 20 mole percent cyclohexene and 80 mole percent toluene? Show your Work on the plot. 1b. Estimate the composition of the distillate of 20 mole percent of cyclohexane if the Distillation column had three theoretical plates.Bristol Community College Fall River, Massachusetts Experiment 6: Molar Mass of a Molecular Solid from Freezing Point-Depression Measurement Name: __________________________________ Date: _______________ Approved: ___________ DATA SHEET Mass of lauric acid (in Part II) Mass of benzoic acid (in Part II) Freezing temperature of pure lauric acid (from Part I) data from Video 2 of Part I Freezing temperature of solution (from Part II) data from Video referenced in Part II Freezing point depression, Tf ( = Tf, lauric acid – Tf, solution) Molality (m) of solution ( Eq. 1) Moles of benzoic acid ( Eq. 2) Experimental molar mass of benzoic acid (Eq. 3) Calculate the molar mass of benzoic acid, C6H5COOH. Percent error Summary Questions A student determines…Assuming that pentane and hexane form an ideal solution, A. Calculate the amount of pentane to be added to 65.0 g of hexane to obtain a minimum dG of mixing from mixing those two components. [MM pentane; 72.15, MM hexane; -86.18] B. Calculate the dS of mixing the components of the mixture above.
- The partial molar volumes of acetone and chloroform, in a solution in which the molar fraction of chloroform is 0.4693, are 74.166 cm3mol-1 and 80.235 cm3 mol-1, respectively. What is the volume of 1,000 kg of this solution? Data: M.M. (acetone) = 58.08 gmol-1; M.M. (chloroform) = 119.07 gmol-1.1. What is the mole percent of methanol in your starting solution (composed of 1.5 mL and 6 mL isopropanol)? 2. Using the vapor-liquid equilibrium data graph (NOTE: You have to make this graph using the data provided) and your answer from question 1, answer the following: If your first distillate was 49 mole percent methanol, how many theoretical plates were in your distillation setup? Data Mole % of Methanol Temperature (°C) Vapor Liquid 66.22 95.35 90.10 67.94 89.10 79.00 70.22 80.00 66.05 72.67 68.50 52.20 74.78 57.00 40.80 77.06 42.85 29.30 78.94 29.00 19.50 81.00 13.20 8.10Given that a mixture of nitric acid (bp 86 °C) and water forms a maximum-boiling-point azeotrope that boils at 120.7 °C with a composition of 67.4% nitric acid to 32.6% water. Construct an approximate boiling-point-phase diagram (with % composition on the x-axis and temperature on the y-axis) for this system. Include the boiling points of pure nitric acid, pure water, and the boiling point of the azeotrope on your diagram. Describe the behavior on distillation of a mixture that is 80% water and 20% nitric acid.
- There are two separate phases, one with a composition of 12% phenol and the other with a composition of 60% phenol, from a 210 gram phenol-water mixture at 60 °C with a composition of 35% phenol. Calculate the masses of these phases.There are two separate phases, one with a composition of 12% phenol and the other with a composition of 60% phenol, from a 210 gram phenol-water mixture with 35% phenol at 60oC. Calculate the masses of these phases.Using the phase diagram for mixtures of cyclohexane and toluene, estimate: A) Boiling point of pure toluene _________ B) Boiling point (Tbp) of solution with molar fraction of toluene 30%.______ C) Molar fraction of toluene (in %) in the gas phase above the solution with molar fraction of toluene 30% at its boiling point Tbp. ____________ D) Approximate molar fraction of solvent which remains in the liquid phase upon heating of mixture with 60% of toluene to 100oC. _______________
- I’ve done this question but I think I’ve done it wrong. Calculate how much 95% Ethel alcohol will be prepared to dissolve 0.3g of sulfanilamide at 78 degrees using the table (pictured below from my textbook) and, based on that volume, calculate how much sulfanilamide will remain dissolved in the mother liquor after the mixture is cooled to 0 degrees. For the volume at 78 I got 1.47mL and for the difference once it cooled to 0 degrees I got 19.96 mL. If I got these wrong could you point me in the right direction as how to solve this? Thank you!3. Refer to the phase diagram depicted below: When 7 moles of A and 3 moles of B are mixed at 50oC which is then cooled to 10oC the sample turns into two liquid phases, phase I has mole fraction xB=0.14 (see diagram below). Find the phase II mole fraction xB, and then calculate the ratio of amount of substance in phase I to that in phase II using Lever rule. Please enter your answer as a number with two decimals. for example, if the ratio you calculated is 2:3, you enter as 0.67The experimental data in the table was collected during a freezing point depression study where BHT (butylated hydroxytoluene) was the solvent. Mass of BHT Mass of unknown Freezing point of pure BHT Freezing point of BHT and unknown solution Kf for BHT 7.709 g7.709 g 1.252 g1.252 g 74.17 ∘C74.17 ∘C 70.91 ∘C70.91 ∘C 6.83 ∘C/?6.83 ∘C/m Use this data to calculate the molar mass of the unknown solute. molar mass =

