6. What are the units for the rate constant, k, in this reaction? Are rate constant units always the same for every different reaction? Explain. 7. It was mentioned in the handout that what we measure in this experiment is the (average) initial rate of the reaction. We measure the time it takes for the first 6.0 x 104 M of I3 to form, and get the rate by dividing the change in concentration (=6.0x104 M) by that time. After this, the reaction then continues. What happens to the rate? Explain your answer. 8. Why do you think we say that you should stop timing at "the first appearance of blue color"? (Think about the consequences if some people stopped at the first appearance, and others waited until the flask was totally blue.)
6. What are the units for the rate constant, k, in this reaction? Are rate constant units always the same for every different reaction? Explain. 7. It was mentioned in the handout that what we measure in this experiment is the (average) initial rate of the reaction. We measure the time it takes for the first 6.0 x 104 M of I3 to form, and get the rate by dividing the change in concentration (=6.0x104 M) by that time. After this, the reaction then continues. What happens to the rate? Explain your answer. 8. Why do you think we say that you should stop timing at "the first appearance of blue color"? (Think about the consequences if some people stopped at the first appearance, and others waited until the flask was totally blue.)
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
100%
Answer questions 6,7 and 8 please
Answer ASAP PLEASE
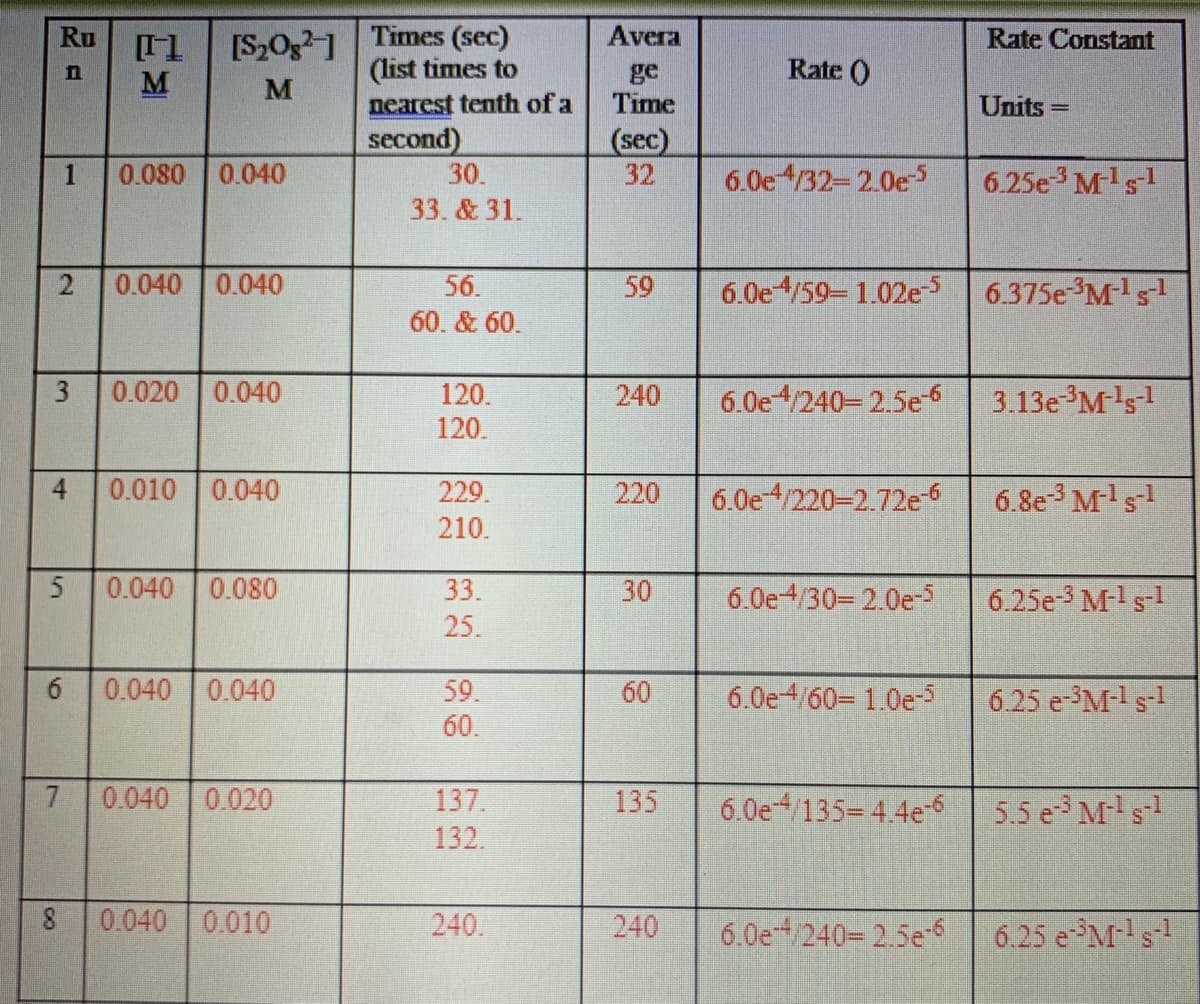
Transcribed Image Text:[S2Og1 Times (sec)
M
Ru
Avera
Rate Constant
(list times to
Rate ()
ge
Time
M
nearest tenth of a
Units
%3D
second)
(sec)
32
1
0.080 0.040
30.
6.0e4/32-2.0e5
6.25e3 Mls
33. & 31.
0.040 0.040
56.
59
6.0e4/59 1.02e
6.375e M-s
60. & 60.
3
0.020 0.040
120.
120.
3.13e M's1
240
6.0e4/240-2.5e
4
0.010
0.040
229.
220
6.0e4/220-2.72e6
6.8e M's
210.
5
0.040 0.080
33.
30
6.0e4/30= 2.0e-
6.25e-3 M-ls-l
25.
0.040 0.040
59.
60
6.0e4/60- 1.0e-5
6.25 e-3M-1 s-1
60.
7.
0.040 0.020
137.
135
6.0e /135= 4.4e-6
5.5 e Ms1
132.
0.040 0.010
240.
240
6.0e /240= 2.5e6
6.25 eM-s-!

Transcribed Image Text:3. Why is it more instructive in detemining reaction order, to compare the rate of Expt 4 to Expt 3 rather than
comparing Expt 3 to Expt 5?
4. Which experiment(s) had the greatest rate? Why was this the greatest rate? Which had the smallest rate?
Why was this the smallest rate? Can you make a general statement about what rate depends on from the
answers to this question?
5. Compare the value of k (the rate constant) for each of the experiments. Is it similar, the same, differet?
It that what you expected? Why is it called a constant?
6. What are the units for the rate constant, k, in this reaction?
Are rate constant units always the same for every different reaction? Explain.
7. It was mentioned in the handout that what we measure in this experiment is the (average) initial rate of the
reaction. We measure the time it takes for the first 6.0 x 104 M of I3 to form, and get the rate by dividing
the change in concentration (=6.0x104 M) by that time. After this, the reaction then continues. What
happens to the rate? Explain your answer.
8. Why do you think we say that you should stop timing at "the first appearance of blue color"? (Think about
the consequences if some people stopped at the first appearance, and others waited until the flask was totally
blue.)
9. Look at Table 1, where you recorded the data for times of each experiment. Take experiment 1. Look at the
times for that experiment. What are the typical fluctuations in the times? In other words, are the different
time measurements for this experiment within 2 seconds of each other? 5? 10? More? Less? How many
seconds? Do this for experiment 2 as well. Think of this range of seconds as the "error" in the time
measurement for each experiment. Now answer this question... do you think that if you added or subtracted
this "error" in the times from the average times for each experiment that it would alter your conclusion about
the reaction order? Please give this some thought. What does this tell you about the reliability of your
reaction order choices?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
