Q: Calculate the partial pressure (in Torr) of Hg if a 50.40 g sample of nitrogen-mercury vapor at 23°C...
A:
Q: Determine the concentration in mol/L (M) of a solution of 18.25 g of HCl in 250.0 mL of water and id...
A: The molecular weight of HCl is 36.5 gm/mol That 36.5 gm HCl=1mol Hence 18.25 gm HCl=18.25gm×1mol/36....
Q: For the following data set: 18.3g, 184g 13g 15.0S Determines whether the questionable data is retain...
A:
Q: Computation Mefenamic acid 500 mg Lactose 200 mg To make 1 cap Based on the given ingredients and am...
A: Based on the amount of ingredients, size of the capsule will be selected.
Q: Experiment No: 10 SPECTROPHOTOMETRIC DETERMINATION OF IRON RESULTS AND INTERPRETATIONS Table 10.1. M...
A: Given:
Q: Dtermine the formal charge of N and the number of lone pairs present.
A:
Q: Computation. What is the amount in milligrams of white wax needed to prepare 10 grams of white ointm...
A: This problem will be solved by unitary method. Calculations are done on the following step.
Q: Calculate the degree of unsaturation. Assign the relevant peaks in the IR spectrum. Assign the peaks...
A:
Q: 03 CN What acid-base theory is involved here? The conjugate base of which acid is relatively more st...
A: This question is abased on the Bronsted lowry concept. According to which if a compound guves H+ it ...
Q: Calculate the degree of unsaturation. Assign the relevant peaks in the IR spectrum. Assign the peaks...
A: The structure of the following is:
Q: 3. Rank the following in order of increasing acid strength (1-weakest acid, 15 strongest; add rank n...
A:
Q: Calculate the percentage of nitrogen, N, in dinitrogen trioxide, N203. A. 46.7% B. 18.4% C. 36.8% D....
A: The molecular weight of N2O3 is (14×2+16×3)=76 gm/mol Weight of nitrogen in N2O3 is (14×2)=28gm
Q: Using the data in the table, calculate the standard entropy changes for the following reactions at 2...
A:
Q: Which of the following CANNOT be found in a safety data sheet of a chemical? (A Hazard Identificatio...
A: Safety data sheets in the laboratory contains useful informations about the identification of hazard...
Q: Aqueous hydrochloric acid (HCI) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium ...
A:
Q: The molecular formula of a hydrocarbon which has a molecular weight of 84 amu and which contains 85....
A: Molecular weight = 84 amu Mass percentage of C = 85.7 % Molecular formula of a hydrocarbon = ?
Q: An increase in temperature drives an endothermic reaction toward the reactants. True False
A: Temperature effect the equilibrium directions
Q: Codeine can treat pain and cough but has a high risk for addiction and dependence. Which of the foll...
A:
Q: According to Kolrausch’s law of independent ionic migration, the molar conductivity of an electrolyt...
A: We can find the molar conductivity of an electrolyte at infinite dilution with the help of Kolrausch...
Q: etermine the formula weight of Ca 3(PO 4) 2. |a. 310 amu O b.215 amu O c. 279 amu O d. 135 amu D e. ...
A: Given compound Calcium phosphate
Q: n aqueous solution of potassium hydroxide is standardized by titration with a 0.178 M solution of hy...
A: Molarity is defined as number of moles of solute divide by volume of solution
Q: Name of Experiment: Fluorescence Spectroscopy Q1: Purpose of experiment 02: Inttroduction
A: Experiment Name - Fluorescence Spectroscopy 1) purpose of Experiment - It is a widely accepted an...
Q: Cu(OH)2 + HNO3
A:
Q: the infrared region, there is an absorption line of hydrogen that appears at 1094 nm. Determine the ...
A: Ans. This transition is part of The Paschen Series of lines which occurs in the infra-red region of ...
Q: The molar conductivity at infinite dilution of H* and NO3 ions at 298K are 349.8 Scm-mol & 71.44 Scm...
A: According to Kohlrauschs law molar conductivity of an electrolyte at infinite dilution is equal to ...
Q: In the gas phase reaction 2 A(g) + B(g) = 3 C (g) + 2 D (g), it was found that when 1.00 mol A, 2.00...
A:
Q: Fil in the blank with the reagents necessary to complete the synthesis shown Drag the appropriate la...
A:
Q: 1. Circle the correct NET ionic equation for the reaction (if any) that occurs when aqueous solution...
A: A question based on general chemistry that is to be accomplished.
Q: The addition of 3.15 g of Ba(OH)2 · 8H2O to a solution of 1.52 g of NH4SCN in 100 g of water in a ca...
A: As per our guidelines we can only solve first question. Please resubmit the other questions as next ...
Q: Which of the following pairs is correctly matched? A hotplate : supplies flame B beaker : measures v...
A: The options given are,
Q: What is oxidation number?
A: Given question is : What is oxidation number = ?
Q: A 21.35 g sample of a substance is initially at 20.6 °C. After absorbing 2231 J of heat, the tempera...
A:
Q: In a voltammogram, which of the following statements is correct a. The limiting current i, is used f...
A: Among the options given option a) the limiting current used for quantitative analysis is correct . T...
Q: Which of the following is a correct practice in handling reagents? A Return any excess reagent to it...
A: The options given are,
Q: Which of the following pairs is incorrectly matched?
A: Option D
Q: Determine the empirical formulas for compounds with the following percent compositions: 15.8% carbon...
A: Empirical formula is defined as the simplest whole number ratio between the atoms of the molecule.
Q: Isomerisation of [Co(NH3);NO2]Cl2 (Nitro) to [Co(NH;};ONO]CI, has been studied extensively using IR ...
A: The absorbance is measured at 1055 cm-1.
Q: WHICH OF THE FOLLOWNG PHYSICAL PROCESSES IS EXOTHERMIC? A. BOILING OF WATER B. MELTING OF ICE C.E...
A: WHICH OF THE FOLLOWNG PHYSICAL PROCESSES IS EXOTHERMIC? BOILING OF WATER, MELTING OF ICE EVAPORATI...
Q: molar conductivity
A:
Q: Select from each set the element with highest IE1 (matching sets are color-coded): Set (a): Set (b):...
A: Ionization energy is defined as the amount of energy required ro remove electron from a neutral isol...
Q: A chemist obtained the following data for the percent compound Z in triplicates (n=3) of an insectic...
A: Data obtained by the Chemist:- The percent compound Z in triplicates (n=3) of an insecticide prepara...
Q: 22.00 g sample of nitric acid (HNO3) has a purity of 96.20% by mass. Calculate the following: the ...
A:
Q: Experiment: UV-Vis Spectrophotometry and Beer’s Law Absorption of Light of Dyes in Beverages Ex...
A: Spectroscopy is basically a technique that study the interaction of matter with radiation UV spectr...
Q: Question 25 The standard cell potential of the reaction is 1.10 V for the cell Zn (s)/Zn2+//Cu²+/Cu ...
A:
Q: Calculate the heat capacity, in joules per degree, of 28.4 g of water. A) 18.0 J/°C B) 118.7 J/°C c)...
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: An unknown compound has the following chemical formula: PtCly where x stands for a whole number. Mea...
A: Here, the unknown compound given is PtClx. No of moles of Pt and Cl are given .we have to find chemi...
Q: Which of the following pairs is correctly matched? A spatula : scoops out solids B crucible tongs : ...
A: Spatula, crucible tongs, beaker and hotplate are tools used in the laboratory.
Q: 03 CNS Explain acid/base strength of A or B
A: The compound which remove easily H+,this compound is more acidic comparetivly others In case of com...
Q: Consider the reaction below. Given the equilibrium concentration of each species, what is the equili...
A: Equilibrium constant ( Kc ) is the ratio of concentration of products to concentration to reactants ...
Q: Aluminium metal reacts with sulphuric acid, H2SO4, to yield aluminium sulphate and hydrogen gas. Cal...
A: Aluminium metal reacts with sulphuric acid, H2SO4, to yield aluminium sulphate and hydrogen gas. The...
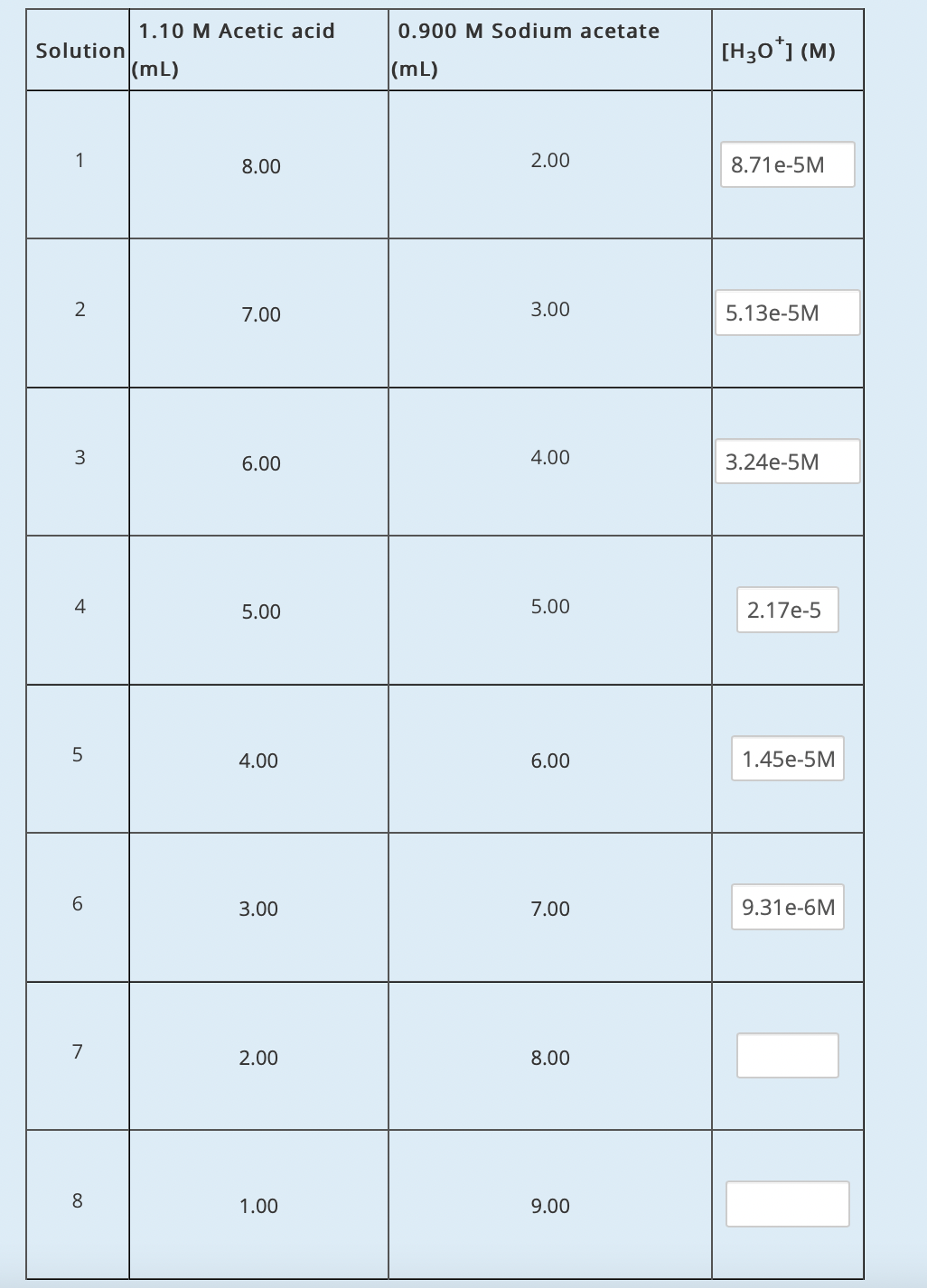
Step by step
Solved in 3 steps with 3 images

- Table 1: Temp vs. solubility data of ammonium chloride. Test tube g NH4Cl/10 mL H2O Crystallization temp. (ºC) 1 1.0 g/10 mL H2O 10 oC 2 1.8 g/10 mL H2O 20 oC 3 2.9 g/10 mL H2O 40 oC 4 3.8 g/10 mL H2O 65 oC 5 4.4 g/10 mL H2O 95 oC Use the table to plot a solubility curve for ammonium chloride on the graph provided below. For the best fit line, use a smooth curve. "Mass of Solute per 100 mL of H2O," should be written on the y-axis (use increments of 2g for every box). "Temperature (°C)" should be written on the x-axis (use increments of 10 °C for every 2 boxes).Concentration of iodide solution (mM) 100.0 Search And Select Concentration of thiosulfate solution (mM) 22.8 Concentration of hydrogen peroxide solution (mM 176.0 Temperature of iodide solution (\deg C) 25.0 Volume of iodide solution (1) used (mL) 10.0 Volume of thiosulfate solution (S2032-) used (mL) 1.0 Volume of DI water used (mL) 2.5 Volume of hydrogen peroxide solution (H2O2) used (mL) 7.5 Time (s) 12.8 What is the Initial concentration of hydrogen peroxide in reaction ( mM)?If 500mL of 0.10M Ca2+ is mixed with 500mL of 0.10M SO42-, what mass of calcium sulfate will precipitate? Ksp for CaSO4 is 2.40 • 10-5. express answer to 3 significant figures. And include units.
- It is known that acid content has a major effect on theflavor of vinegars, but most cheaper vinegars are diluted similarly to 5% acidity Wt./vol. % is equivalent to gsolute per 100mL solution (so 5% is equivalent to 5 g acid/100 mL solution). a.) First, calculate the approximate molar concentration of acetic acid in the 5% wt./vol vinegar. b.) Next, calculate the expected molarity of acetic acid in the solution upon dilution by a factor of 5. Thank you!KNO3 (aq) added to CaC2O4 (Ksp = 1.3 x 10-8) in solution will lead to __________ in solubility of the solid no effect then decrease increase no effectBen prepared a salicylic acid in pentanol solution by adding 4.77 g of salicylic acid to 91.88 mL of pentanol. Calculate the mole fraction of salicylic acid in this solution. MW of salicylic acid= 138.12 g/mol MW of pentanol= 88.15 g/mol Density of pentanol= 0.8144 g/mL
- The following evidence was obtained from an experiment to determine the solubility of calcium chloride at room temperature. A sample of saturated calcium chloride solution was evaporated to dryness, and the mass of solid residue was measured.EvidenceVolume of solution (mL) = 15.0Mass of empty beaker (g) = 90.54Mass of beaker and residue (g) = 101.36The solubility of calcium chloride is g/100 mLMixing 150.0 mL of 1.50 M Ba(NO3)2 (aq) with 250.0 ml of 5x10-2M Na2SO4 at 25°C. The Ksp of barium sulfate is 1.08x10-10M2 at 298 K. What is the final concentration of barium ion in the solution.Calculate either [H3O+][H3O+] or [OH−][OH−] for each of the solutions at 25 °C.25 °C. Solution A: [OH−]=2.25×10−7 MSolution A: [OH−]=2.25×10−7 M Solution A: [H3O+]=Solution A: [H3O+]= MM Solution B: [H3O+]=9.29×10−9 MSolution B: [H3O+]=9.29×10−9 M Solution B: [OH−]=Solution B: [OH−]= MM Solution C: [H3O+]=0.000649 MSolution C: [H3O+]=0.000649 M Solution C: [OH−]=Solution C: [OH−]= M
- Submit a clean, dry, and properly labeled 50-mL reagent bottle for your unknown solution. Pipet 20.00 mL of the sample in 250-mL Erlenmeyer flask. Add 5 mL of buffer and 5 drops of indicator. Titrate the solution until it turns light blue. If the titration consumes more than 50 mL of the titrant, dilute the sample accordingly. Compute for ppm of CaCO3 using the following table. Mean Molarity = 2.487 x 10^-3 MBlank correction = 0.015 Formula = V (mL) titrant x Mean Molarity of Titrant x Molecular weight of CaCO3 x 1/V (L) sampleA solution is prepared by dissolving 40.00 g of NaCl (f.w. = 58.44 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 1 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Find the freezing point of the solution(in C to 2 decimal places)A solution is prepared by dissolving 40.00 g of NaCl (f.w. = 58.44 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 1 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Find the osmotic pressure in atm to three decimal places





