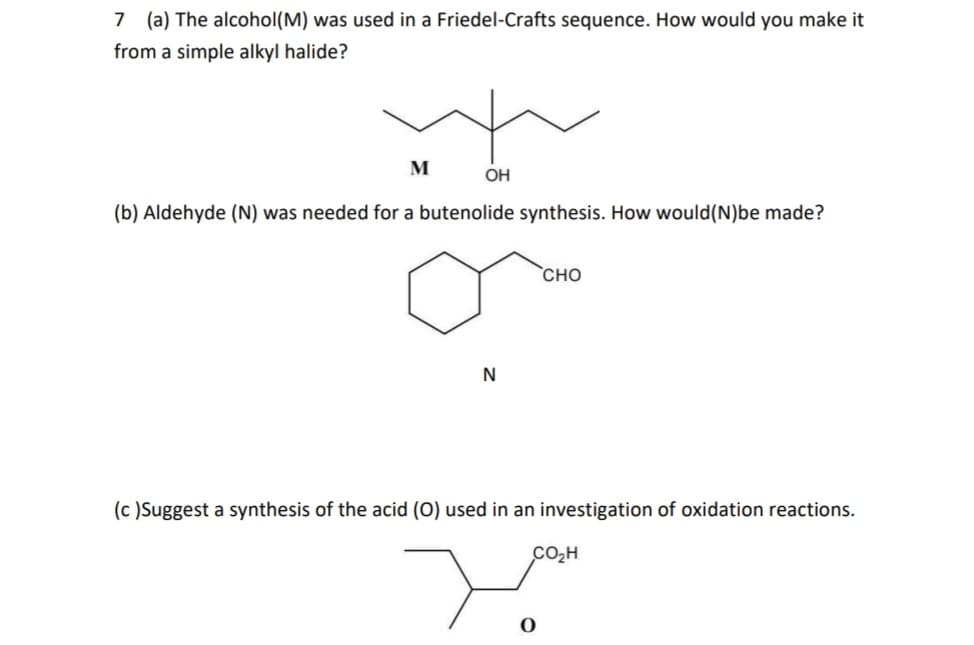
7 (a) The alcohol (M) was used in a Friedel-Crafts sequence. How would you make it from a simple alkyl halide? M OH (b) Aldehyde (N) was needed for a butenolide synthesis. How would (N)be made? N CHO (c) Suggest a synthesis of the acid (O) used in an investigation of oxidation reactions. CO₂H سید
Q: Solve for the equilibrium concentrations of B and C. Be sure each has the correct number of…
A:
Q: What is the percentage of HCl in the solution if the molarity is 0.268398000? Assume the density of…
A:
Q: Identify how the compounds below are related to one another. Select the single best answer.…
A: Answer: (A) EnantiomersExplanation:Answer: (A) Enantiomers: Stereoisomers that are…
Q: A certain first-order reaction is 58% complete in 65 s. What are the values of the rate constant and…
A: k= 1.33 x10-2 s-1 t 1/2 = 52.1 sExplanation:Step 1: Step 2: Step 3: Step 4:
Q: Classify each chemical reaction as endothermic or exothermic. Endothermic reactions Answer Bank…
A: The objective of the question is to classify the given chemical reactions as either endothermic or…
Q: Which structure(s) will have an odd-numbered molecular ion? ☐ ☐ H2N- ΝΗ 'NH2 Ν :0: Ν
A: Molecular ions are created when electrons are added or removed from neutral molecules. A molecular…
Q: Part G Give the IUPAC name for compound shown below. Spell out the full name of the compound.…
A:
Q: Predict the reactants of this chemical reaction. That is, fill in the left side of the chemical…
A: Balanced molecular equation can be define as the reaction in which number of atoms of all the…
Q: 1 atm C 0.006- atm B -25 D A 0.01 25 T 100 A. Aliquid, B = solid, C = gas, D = critical point B. A…
A: Phase diagram:The phase diagram represents the different phases of the substance and the pressure…
Q: What is the relationship between the parent structure on the left and the structure on the right? a…
A: Answer:Compounds with same molecular formula but different structural formulas are called…
Q: Tooth enamel consists of hydroxyapatite, Ca5(PO4)3OH (Ksp = 8 × 10−37). Fluoride ion added to…
A: The objective of this question is to calculate the solubility of hydroxyapatite (Ca5(PO4)3OH) and…
Q: Which option is the correct line-bond structure of the given molecule? CH₂ HC-C-CEC-H CH3 O O O O
A: To find the bond-line structure of the compound given.
Q: TOWS ITTANCE 20:30 3500 Copyright 1994 1H 24 atlaslaste 6.8 6.4 6.2 8.0 Proton NHA B 2010.23 2520…
A: The characteristic bands can be tabulated as; Wavenumber (cm-1)Bondvibration shape intensity…
Q: What is the pH of a solution containing 1.053 mol L-1 of a diprotic acid with PKA1 = 3.93 and pKA2 =…
A: Given data:Determine the pH of the solution of diprotic acid.
Q: If 115 cal are added to 49.2 g of solid iron, what is the change in temperature of the iron? The…
A: The objective of this question is to calculate the change in temperature (ΔT) of the iron when a…
Q: In a study of the formation of HI from its elements, H2(g) + 12(g) = = 2HI(g) equal amounts of H2…
A: Initially, both H2 and I2 concentrations decrease as they react to form HI, so their concentration…
Q: Draw the structure of all products of the mechanism below. :CI: :0: + H₂C-C―OCH₂ Al Click and drag…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: A chemist dissolves 130. mg of pure barium hydroxide in enough water to make up 100. mL of solution.…
A: The values are provided in questionmass of barium hydroxide = 130 mgmass of barium hydroxide = 130…
Q: . The rate of vaporization of a liquid can be increased by 1. increasing the surface area 2.…
A: rate of vaporization, is the ratio of how quickly a material vaporizes compared to another known…
Q: estion 12 of 33 Write down the common (not IUPAC) names of the organic molecules that would be…
A: It is the process of splitting ester bonds with water.
Q: The depletion of ozone in the stratosphere can be summarized by this equation: 203(g) 302(g) 03(g)…
A: The objective of the question is to determine-i) The change in entropy and the change in Gibbs free…
Q: Potassium nitrate, KNO3, has a molar mass of 101.1 g/mol. In a constant-pressure calorimeter, 41.8 g…
A:
Q: 7 With the Polygon in a Circle method, which one has a configuration? ⑦With the shell closed shell
A: Open shell configuration means there is the presence of unpaired electrons, i.e., there are some…
Q: Draw a structural formula for the major organic product(s) of the reaction shown below. 'N' H 1.…
A: These both reactions are good examples of Hoffman exhaustive methylation.
Q: The decomposition of N2O5(g) to NO2(g) and O2(g) obeys first-order kinetics, where k = 3.4 x 10–5…
A: The objective of the question is to calculate the initial rate of reaction for the decomposition of…
Q: Name the molecules & Identify any chiral center CH3CH2CH2CHCH2CH2CH2CH3 OH CH₂CHCH2CH3 Br…
A: Stereogenic centers also called chiral centers in the organic molecule possess four different…
Q: Please draw diagram for step 1, 2 and 3
A: Below is the clarified and detailed explanation for above tricky question.....if you are ok with my…
Q: i:2.5/4.0/0.25/0.4 ii:6.25/75/25/100/50 iii:25/50/75 iv1:4/0.2/0.75/0.25 iv2:8/10/20/30/40
A: 1. 0.252. 100ml3. 75ml4a. 0.24b. 8%Explanation:
Q: A2B A2BA AB AB2 Number of Electrons Given the data below, if atoms of A would react with atoms of B,…
A: The molecular formula will be: AB2Explanation:Thank You.
Q: Which of the following lipids will have the lowest melting point? HO о O:
A: These are the fatty acids or their derivatives which are insoluble in water but can be soluble in…
Q: Carboxylic acids can be made by the hydrolysis of nitriles, which in turn can be made from an alkyl…
A: Alkyl bromides are a good substrate for nucleophilic substitution reactions. If an alkyl bromide…
Q: % Leaving Group, Tertiary Sketch and Submit Above Carbocation, NOS neutral nucleophile Addition…
A: During a nucleophilic substitution reaction, a nucleophile substitutes an atom or group of atoms…
Q: 0.5 PH3 P4 H3PO₂ 8. The following is a Frost diagram of phosphorus in aqueous solution at pH = 0.…
A: The objective of the question is to identify the-Most Thermodynamically Stable FormLeast…
Q: b) OH OCH, d) H 1) CH3MgBr 2) H₂O* 1) SOCI₂, heat 2) (CH3CH2)2CuLi 3) H₂O 1)2 eq. CH3CH2MgBr 2) H₂O*…
A:
Q: Give the sublevel designation, the allowable ml values, and the number of orbitals for the…
A: The objective of the question is to determine the sublevel designation, the allowable ml values, and…
Q: The reaction A → products is second order. If the initial concentration of A is 0.436 M and,…
A: see attachmentExplanation:Step 1: Step 2: Step 3: Step 4:
Q: Question 18 of 27 Choose the structure that has the formal charge correctly assigned. A) I II) H H…
A: Lewis structure is a method of representing bonding pattern in a molecule.It tells us how different…
Q: 6. H&C. CH2CH3 mild oxidation HO CH3
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: OH OH PCC OH CH2Cl2
A: PCC is a mild oxidising agent and it oxidised primary alcohol to aldehyde and secondary alcohol to…
Q: Balance the following gas-phase reaction and write its reaction quotient, Qc: SClF5 (g) + H2(g) ⇌…
A: We have to balance the following gas-phase reaction and write its reaction quotient, Qc: SClF5 (g) +…
Q: Pure iron, Fe, can be produced from an ore called hematite, Fe2O3, by reaction with carbon at high…
A: In the given reaction->weight of iron = 42.0 tonsThe complete reaction is ->>2Fe2O3 +…
Q: 15. Treatment of the compound shown below with excess m-CPBA (a peroxyacid, RCO3H) in the presence…
A: This is oxidation reaction. MCPBA ,at first there occurs baeyer villiger oxidation in which ketone…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Assume that the water side…
A: Carbonyl compounds (aldehyde and ketone) react with an alcohol to form hemiacetal in the presence of…
Q: CE 3500 Copyright 1994 300 2500 1501.53 2H 3H 2H zi mat 91-1221 254 740.55 Эн порнороско 8.4 8.2 9.0…
A: The characteristic bands can be tabulated as; Wavenumber (cm-1)Bondvibration shape intensity…
Q: Which is the best description of the first law of thermodynamics? ☐ In systems where only the…
A: The objective of the question is to find the best description of the first law of thermodynamics…
Q: A solution with a volume of 1.00 L is 0.550 M in CH3COOH(aq) and 0.450 M in CH3COONa(aq). What will…
A: Answer:Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: Which one is antiaromatic? (c) a) b) auti D e) not sp3 from aromatic
A: Aromatic compound - obey huckel rule and contains 6 π eAnti aromatic compound - don't obey huckel…
Q: Assign the "H-NMR peaks belonging to the structure of the Grignard product and explain. HB HA -OH 9…
A: The assignment of the protons in the given compound is shown as follows:#types of protons =…
Q: Need for CLF3. CHECK OTHER IMAGE FOR SAMPLES I NEED LIKE THIS SOLUTIONS OK. OTHER IMAGE I NEED…
A: Lewis structure shows the relative distribution of bond pairs and lone pairs present in the…
Q: he cloth shroud from around a mummy is found to have a 14C activity of 9.3 disintegrations per…
A:
Step by step
Solved in 1 steps with 1 images

- 1.Describe the ozonolysis of alkenes 2.one mole of a hydrocarbon(A) reacts with one mole of beomine giving a dibromo compound C5H10Br2.Substance A on treatment with cold dilute kMnO4 solution forms a compound C5C12O2(C5H12O2) on ozonolysis A,gives equimolar quantities of propanone and ethanol.Deduce the structure of substance A.A. In the synthesis of 1-bromobutane, what is the inorganic by-product left in the reaction flask following the distillation? Why was the bromoalkane the bottom layer in the separatory funnel? B. Predict the product when 1-methylcyclohexanol reacts with H2SO4 and KBr. Show the mechanism.3. Benzalacetophenone can be nitrated to give the mononitro derivative.(a) Provide the structure of the mononitro derivative.(b) If a second nitro group is introduced, where would it enter? Give the structure of theproduct.
- Explain how benzaldehyde and dimedone reacts with each other, and then with the aminotriazole to form compound 1a in the presence of an acid catalyst. Provide a detailed reaction mechanism. During the development of the optimized procedure for the experiment, it was found out that compound 1b can also be produced from the same set of starting materials. Propose a detailed reaction mechanism for the formation of 1b. Explain your answer. What factor/s may drive the formation of 1b over 1a?Propose a feasible synthesis for 2-iodo-4-methylphenolfrom2-amino-4-methylphenol. Draw the reaction scheme and briefly describe the reaction.(a) Explain the mechanism of a nucleophilic attack on the carbonyl group of an aldehyde or a ketone.(b) An organic compound (A) (molecular formula CgH16Q2) was hydrolysed with dilute sulphuric acid to give a carboxylic acid (B) and an alcohol (C). Oxidation of (C) with chromic acid also produced (B). On dehydration (C) gives but-1-ene. Write the equations for the reactions involved.
- Doxaprost, an orally active bronchodilator patterned after the natural prostaglandins , is synthesized in the following series of reactions starting with ethyl 2-oxocyclopentanecarboxylate. Except for the Nef reaction in Step 8, we have seen examples of all other types of reactions involved in this synthesis. Q. Write equations to show that Step 6 can be brought about using either methanol or diazomethane (CH2N2) as a source of the -CH3 in the methyl ester.a) Give the appropriate base for the above reaction and draw the resonance structures of the enolate ions derived from compound A. Then write the mechanism for the formation of the Dieckmann cyclized product B. b) If compound B above is reacted with NaBH 4 , draw the structure of the reduced product. Give a reason for your choice of product. c) Being a β-ketoester, B could undergo a 3-step synthesis involving alkylation, hydrolysis and decarboxylation reactions to yield the cyclopentanone, C. Write the outline synthesis for each step which include the appropriate regents and correct intermediate.Propose a mechanism for the reaction of aniline with ethyl acetate to give acetanilide.What is the leaving group in your proposed mechanism? Would this be a suitableleaving group for an SN2 reaction?
- Sketch and explain the IR, UV, mass, and NMR (1H and 13C) spectroscopy data of BenzonitrileSuggest reagents to make the drug phenaglycodol by the route below.Treatment of salicylaldehyde (2-hydroxybenzaldehyde) with bromine in glacial acetic acid at 0°C gives a compound with the molecular formula C7H4Br2O2, which is used as a topical fungicide and antibacterial agent. Propose a structural formula for this compound

