7 Use the References to access important values if needed for this question. According to the following reaction, how many moles of oxygen gas are necessary to form 0.963 moles carbon dioxide? carbon monoxide(g) + oxygen(g) → carbon dioxide (g) mol Amount = A R Submit Answer 888 % 5 T Retry Entire Group 9 more group attempts remaining 6 MacBook Air Y TAO - 7 U 8 DII ( 9 DD FO O ) O lo F10 P Previous AL F11 { + [ = Next> Save and Exit 642 delete 1 1
7 Use the References to access important values if needed for this question. According to the following reaction, how many moles of oxygen gas are necessary to form 0.963 moles carbon dioxide? carbon monoxide(g) + oxygen(g) → carbon dioxide (g) mol Amount = A R Submit Answer 888 % 5 T Retry Entire Group 9 more group attempts remaining 6 MacBook Air Y TAO - 7 U 8 DII ( 9 DD FO O ) O lo F10 P Previous AL F11 { + [ = Next> Save and Exit 642 delete 1 1
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter9: Chemical Quantities
Section: Chapter Questions
Problem 6QAP: or each of the following reactions, give the balanced chemical equation for the reaction and state...
Related questions
Question
100%
13
Transcribed Image Text:1
Amount =
56
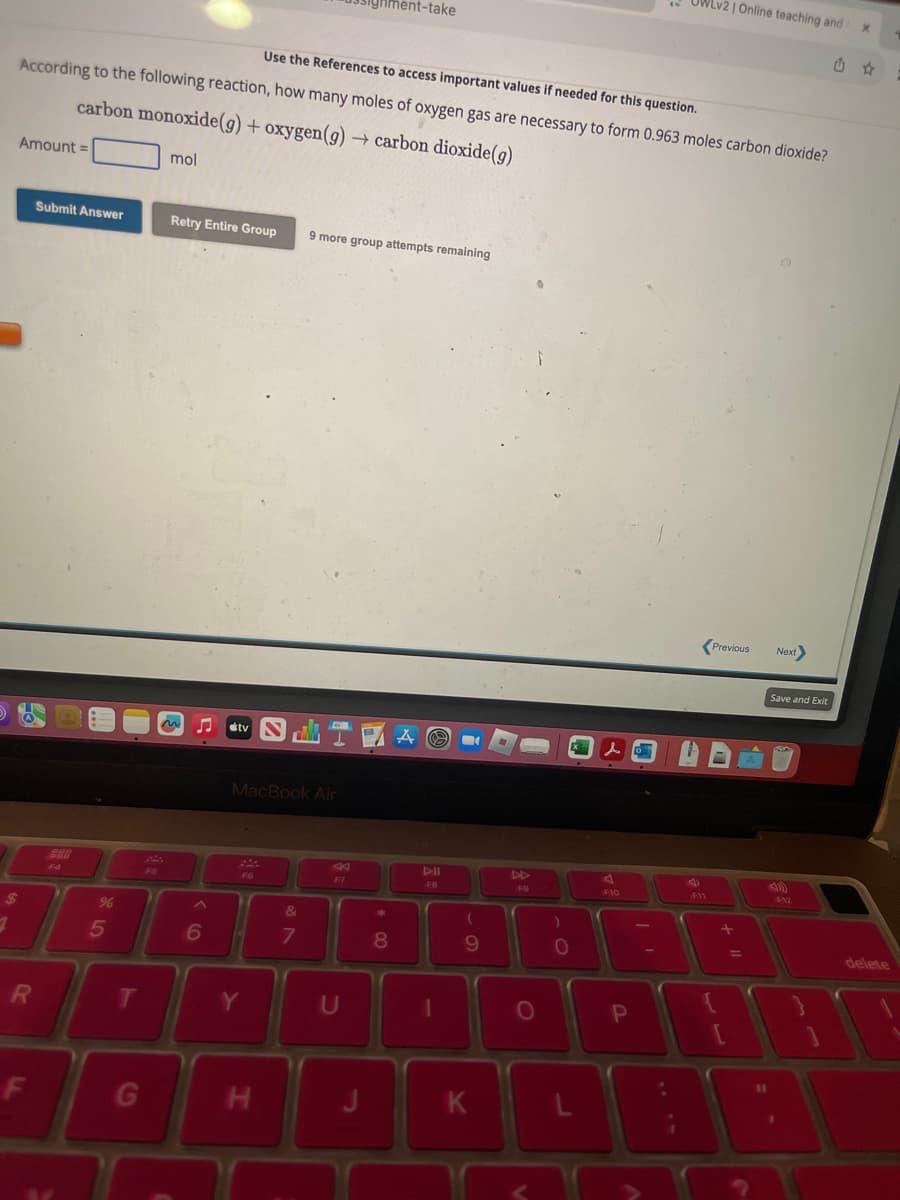
Use the References to access important values if needed for this question.
According to the following reaction, how many moles of oxygen gas are necessary to form 0.963 moles carbon dioxide?
carbon monoxide(g) + oxygen(g) → carbon dioxide(g)
R
F
Submit Answer
%
5
T
G
mol
Retry Entire Group 9 more group attempts remaining
6
MacBook Air
F6
Y
H
ment-take
&
7
U
*
8
A
FB
(
9
K
F9
O
0
d
F10
P
UWLv2 | Online teaching and X
-
F11
Previous
+11
[
Next>
7
Save and Exit
F12
D8B
delete
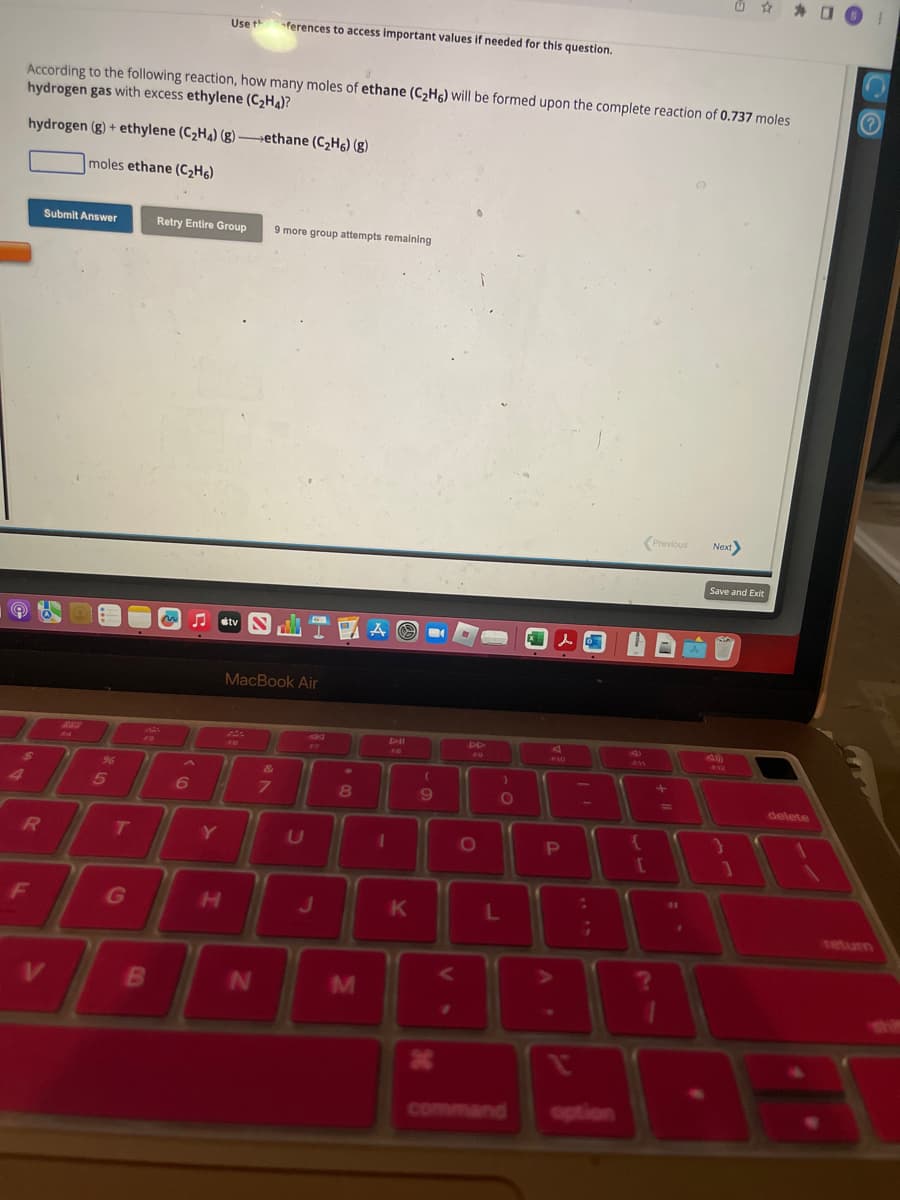
Transcribed Image Text:According to the following reaction, how many moles of ethane (C₂H6) will be formed upon the complete reaction of 0.737 moles
hydrogen gas with excess ethylene (C₂H4)?
hydrogen (g) + ethylene (C₂H4) (g)-ethane (C₂H6) (g)
moles ethane (C₂H6)
e 2
$
R
Submit Answer
F
F4
96
5
T
G
B
Retry Entire Group
6
Use th
Y
H
tv S
ferences to access important values if needed for this question.
N
&
7
9 more group attempts remaining
MacBook Air
www
U
FZ
J
8
M
A
1
D-HI
K
(
9
X
VS
DD
O
)
O
L
lo
P
:
2
¡
?
{
[
Previous
?
DA
+
1
Next>
Save and Exit
F12
1
delete
O O 1
?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning