7. A student gets the following results after running a TLC plate of several over the counter analgesics. What should the student do to get better results? O Run the TLC for a longer time. O Co-spot the sample several times. O Run a new TLC plate in a less polar solvent. O Run a new TLC in a more polar solvent mixture.
7. A student gets the following results after running a TLC plate of several over the counter analgesics. What should the student do to get better results? O Run the TLC for a longer time. O Co-spot the sample several times. O Run a new TLC plate in a less polar solvent. O Run a new TLC in a more polar solvent mixture.
Chapter5: Chromatography
Section: Chapter Questions
Problem 3Q
Related questions
Question
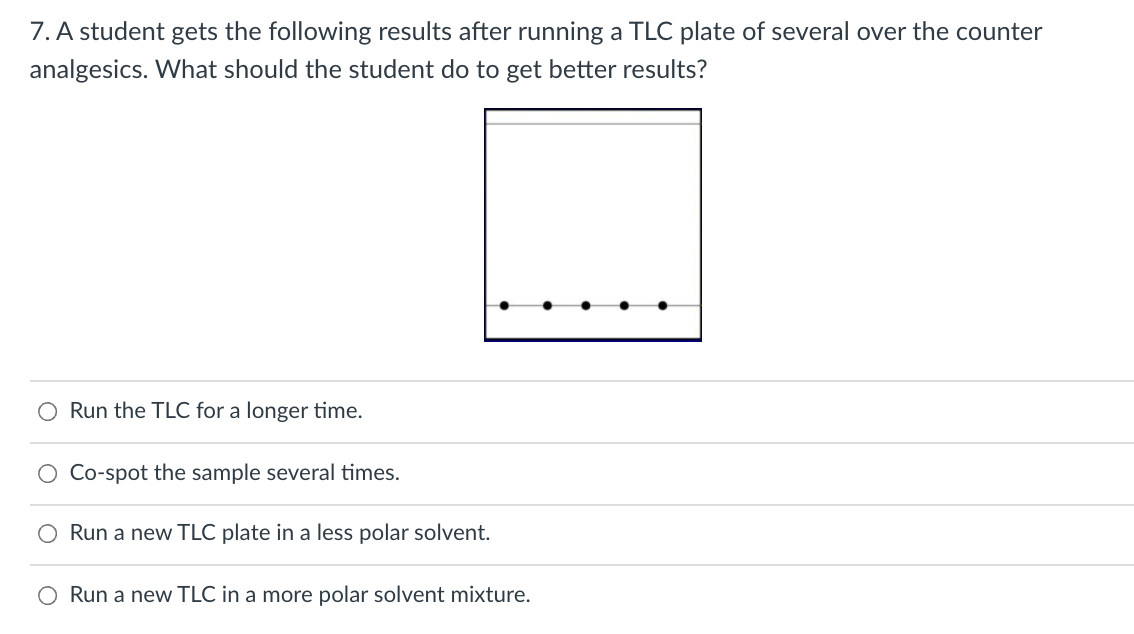
Transcribed Image Text:7. A student gets the following results after running a TLC plate of several over the counter
analgesics. What should the student do to get better results?
O Run the TLC for a longer time.
O Co-spot the sample several times.
Run a new TLC plate in a less polar solvent.
O Run a new TLC in a more polar solvent mixture.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning