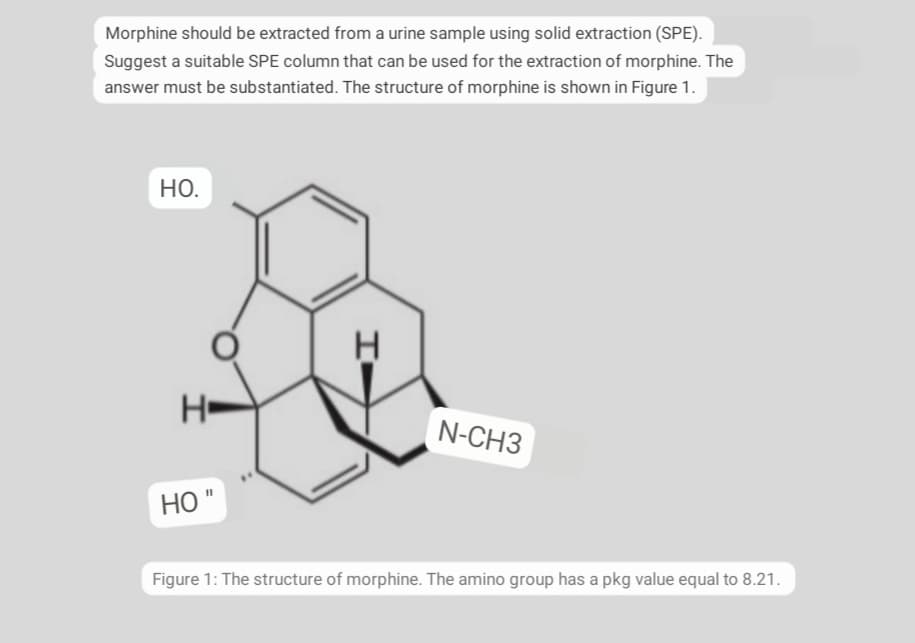
Morphine should be extracted from a urine sample using solid extraction (SPE). Suggest a suitable SPE column that can be used for the extraction of morphine. The answer must be substantiated. The structure of morphine is shown in Figure 1. НО. N-CH3 НО "
Q: A compound contains only C, H, and N. Combustion of 35.0 mg of the compound produces 51.3 mg CO2 and...
A:
Q: Which carbon of the open-chain form of a sugar becomes the anomeric carbon in the cyclic form? ...
A: when a pair of stereoisomer is developed due to the difference in configuration at C1 chiral centre...
Q: U-235 collided with a neutron resulting to a nuclear fission producing La-145, Br-88, neutrons and e...
A: Given data :-
Q: HO H. NH2 H. HO, H2N Cephradine Cephalexin
A: R-S nomenclature: R-S nomenclature is used to determine the stereochemistry of chiral carbons of a m...
Q: a chemist adds 250.0 mL of a 0.053 mM sodium nitrate (NaNO, solution to a reaction flask. Calculate ...
A:
Q: If 5 moles of age are added to 5 moles of O2, what is the limiting reactant?
A: If 5 moles of Ag are added to 5 moles of O2 the limiting reactant is given below.
Q: Acrylonitrile (C3 H3N) is the starting material for many synthetic carpets and fabrics. It is produc...
A: First we have to determine the limiting reagent. Limiting reagent is that which consumed first or fi...
Q: Use the References to access important values if needed for this question. cording to the following ...
A: Given benzene in excess amount
Q: It is a number that use to keep track of how many electrons an atom has? Select one: a. formal...
A: The answer is given below
Q: compound C?
A:
Q: tion 48 Vhich structure is a portion of 1. chitin 2. glycogen 3. Dextran 4. Cellulose? aft4) bond A ...
A:
Q: The following reaction has an average rate of disappearance of C2H5NO2 of 0.02005 M min 1 in the fir...
A:
Q: A chemist must prepare 700. mL of 17.0 µM aqueous zinc oxalate (ZnC,0,) working solution. She'll do ...
A:
Q: Describe at least two methods used to measure pH.
A: Methods of measuring pH.
Q: )0.0100 M in HCl and 0.0320 M in benzoic a ) 0.0100 M in NaOH and 0.075 M in Na,CO )0.0100 M in NaOH...
A: We are given different solutions of mixtures of acids in acids ( part a and b) and mixtures of bases...
Q: Question 2 The following reaction has an average rate of disappearance of H2 of 0.00135 M min in the...
A:
Q: tö access important values if needed for this question. When the following molecular equation is bal...
A: In order to write molecular equation we have to first write reaction.
Q: What number of Ti atoms and what amount (moles) of Ti atoms are in 700.0 g of titanium? atoms Ti mol...
A: Given: Mass of titanium = 700.0 g.
Q: Which of the following statements concerning sugar polymers is false?
A: Answer: In the case of sugar polymers, the branched polymers are more water-soluble than linear poly...
Q: I. Define the following terms: g. Cream h. Lotion i. Cleansing and cold cream
A: A colloid is a mixture is two or more substances mixed together but not chemically combined They ar...
Q: A certain gas is present in a 13.0 L cylinder at 4.0 atm pressure. If the pressure is increased to 8...
A:
Q: For each reaction in the table below, write the chemical formulae of any reactants that will be oxid...
A: The chemical species is oxidized when it adds oxygen or removes hydrogens or removes electrons. The ...
Q: A 0.9550 g sample of ASA was dissolved in 40.1 mL of standard NaOH. The excess NaOH was neutralized ...
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for you...
Q: For nonideal solution, like ethanol and water the less volatile component in this mixture has a ____...
A: Ideal solution is the solution which follows Roult's law and non ideal solution is the solution whic...
Q: Calculate the pH value of the resulting solution by titrating 0.1 molar ammonium hydroxide 100 ml wi...
A: Given that, 100 mL of 0.1 M ammonium hydroxide was titrated with 0.1 M of HCl. The Kb of ammonium hy...
Q: In a solutions, it is a substance that is usually present in greatest amount is known as? Select on...
A:
Q: ionization equation
A:
Q: Question 13 Refer to the schematic diagram below, what is compound D? Conc. H,SO, a-naphthol (-) (*)...
A: A question based in introduction to organic chemistry that is to be accomplished.
Q: An unknown compound has the following chemical formula: Mn(CN) sub x where x stands for a whole numb...
A:
Q: pose 5.056 grams of an unknown solute are dissolved in 50.0 ams of phenol (C6H50H). The resulting s...
A:
Q: What is a unit cell? Sketch a unit cell and label the dimensions a, b, and c and the angles 6. alpha...
A: The smallest repeating unit of the crystal lattice is the unit cell, the building block of a crystal...
Q: Write soluble or insoluble and identify the nature of the solutes on the given solvent: Solute
A: The basic concept behind solubility is like dissolves like, i.e., polar solvents readily dissolves p...
Q: Hi! Can you help me with these? Assignment: Balance these problems. Show your solution. (kindly re...
A: For balancing the reaction, we must need to follow one thing that is number of atoms in L.H.S is equ...
Q: Calculate the molar solubility of yttrium iodate in a 0.0500M solution of magnesium iodate, Mg(IO3)2...
A:
Q: If a strong acid is added in a buffered solution, the hydronium ions react with the conjugate base/s...
A: Consider a buffer solution of weak acid (HA) and A-(salt).
Q: Predicting acid or base strength from the conjugate relative pH of 0.1 M aqueous solution species I0...
A: Answer: In this problem we have to arrange the solution in order of increasing pH.
Q: Gaseous oxygen and carbon dioxide are placed in a sealed flask. After several minutes, the gases are...
A: Since it is is a property of gas that it forms always homogenous solution that is why here both gas...
Q: 2. Identify the kinetic and thermodynamic products of the following reaction. CI HCI (1 еq) Draw a d...
A:
Q: what is compound C
A: In first step give positive test of carbohydrate by treatment with Alpha-naphthanol/H2SO4. Now perfo...
Q: 300.0 mL of a 0.450 M solution of NaI is diluted to 700.0 mL. What is the new concentration of the s...
A: Given that, M1=0.450 M , V1=300.0 mL M2=? , V2=700.0 mL
Q: nes b) ethers
A: Isomers are the compounds which have same chemical formulae but have different arrangement of atoms ...
Q: The Henderson-Hasselbalch equation is formulated based on the ionization of a weak acid O True O Fal...
A:
Q: What kind or type of isomers are the compounds given below? Arrange them in the order of increasing ...
A:
Q: Hemoglobin is a large molecule that carries oxygen in human blood. A water solution that contains 0....
A: Osmotic pressure = concentration × gas constant× temperature...
Q: Higher Rf value will be obtained from dextrin than that of glucose in a reverse-phase TLC. O True O ...
A: Rf means Retardation factor. Rf value means the ratio of distance travelled by solute to the distan...
Q: Calculate osmotic pressure for 0.10 M Na3PO4 at 20°C
A: Answer: Osmotic pressure is the pressure, that is required to be applied on the solution side stop t...
Q: What is the hydronium-ion concentration of a 5.58 x 104 M solution of p-bromobenzoic acid, BrCg H4 C...
A:
Q: buffer solution is formed by adding 0.320 moles of solid sodium lactate, NaC₃H₅O₃, to 600.0 ml of 0....
A:
Q: of red algae. The sample was hydrolyzed and monosaccharide units were analyzed in different colorime...
A:
Q: OH H- H- OH HO-C-H H -C- H-C-H OH Which of the following figures represents the Haworth projection o...
A:
Step by step
Solved in 2 steps

- How much of the suspending agent is needed to prepare a 60-mL preparation of Aluminum hydroxide gel based on this formulation? Ammonium alum 800 g Sodium carbonate 1000g Peppermint Oil 0.01% (0.01 g/100mL) Sodium benzoate 0.1% (0.1 g/100mL) Purified water, qs ad. 2000 mL a. 0.06 g b. 30 g c. 24 g d. 0.006 gYou are given 150 mL of an oil made up of a mixture of 3 fatty acids: palmitoleic acid (C 16:1 (n-7)) erucic acid (C 22:1 (n-9)) stearidonic acid (C 18:4 (n-3)) You know that the 150 mL sample of this oil was prepared by adding 60 mL of palmitoleic acid to 90 mL of a mixture of erucic acid and stearidonic acid. Using a hydrometer you carefully measure the density of the 150 mL sample of oil and your analysis indicates that it has a density of 0.900 g/cm3. The densities of the individual fatty acids are: palmitoleic acid = 0.894 g/cm3 erucic acid = 0.860 g/cm3 stearidonic acid = 0.9334 g/cm3 Can you determine the volumes (in mL) of erucic acid and stearidonic acid in the total volume (150 mL) of the oil mixture?Rank the amino acids used in the experiment in Part II in the order of increasing solubility in water(mobile phase) Phe(0.4), Ala(0.5), Gly(0.8), Ser (0.9), Lys (0.6) and Asp(0.8). The more soluble travels faster.
- purification of egg yolk vitelline 1) Measure the volumes of 3 egg yolks and mix by adding an equal volume of NaCl solution. 2) Extract the mixture with 3 solid volumes of ether and separate the aqueous phase. 3) Do the same procedure 3 times. 4) Mix the sample with water and rinse. 5) It is expected for the protein to collapse. 6) Some more water is added in order to check whether the collapse has occurred completely. 7) The sample is centrifuged and the precipitate is dried.For each step, explain why that action was taken. for example why is NaCl added? Why is the procedure repeated 3 times?what is a reason why company leaders would add melamine to their product to make the protein content appear higher than the actual amount of the product? Describe two possible reasons for why food safety inspectors often use an overall nitrogen content test, instead of Bradford assay, to determine the protein content in a product.Consider the following data that was collected for a protein that contain 0.76 % tryptophan amino acids by weight (Molecular weight of tryptophan amino acids is 204 g/mol). Absorbance Concentration (mg/L) 0.0250 0.100 0.150 0.226 0.259 0.400 0.600 0.964 1.14 1.78 1.45 2.35 1.78 2.87 How many mg of the protein present in 1.00 L solution that shows an absorbance of 1.20?
- One mL of saliva was diluted to 5 mL using distilled water.A 2.00 mL aliquot was obtained and added to 10 mL of 1% starch solution. Form this solution, 0.5 mL was taken and diluted to 5 mL. Calculate the dilution factor.For the amino acid alanine, the major species in solution at pH 7 is the zwitterionic form, which has a negatively charged carboxylic acid group and a positively charged amino group. There is a less common neutral form in which neither group is charged. The carboxylic acid group of alanine has a p?a of 3 The amino group of alanine has a p?a of 8 Estimate the ratio of the concentration of the neutral amino acid species to the zwitterionic species at pH 7. __?__x10^?Explain why casein precipitates when vinegar is added. Why was milk or raw egg once used as an antidote in cases of heavy metal poisoning? Based on your isolated yield of casein, how many grams of the protein are in one 200 g glass of milk? Which of the solutions contained amide bonds? Which test tells you this? Which of the solutions contained compounds with primary amino groups? Which test tells you this? Describe two examples of protein folding patterns and identify what level of protein structure they belong to. What other reagents might you use besides ferric chloride to test affinity for metal ions?
- this is the gc from an experiment we did for the extraction of eugenol from clove oil. i just need the chemical compound associated with each peak labeled. thank you. A flowchart of the experiment is included as well. Thank you.Create a Schematic Diagram Delivering an aliquot and dilution1. Using a serological pipette, transfer accurately a 10-ml aliquot of the previously prepared NaCl solution into a 100-ml volumetric flask, then dilute to mark using distilled water.2. Put a stopper on the volumetric flask. Then invert the glassware up and down around five times, making sure there are no leaks.3. Put the diluted solution in a reagent bottle, then properly label the solution. Note the concentration of the solution. Show your reagent bottle to your instructor for checking.Erwin Smith wants to determine the average concentration of amino acids present in the spinal fluid of Eldians. He assigned four people to complete this task. Who among these four people will be able to obtain the best results? a.Levi Ackerman, who obtained samples from 100 Eldians from Wall Maria b.Mikasa Ackerman, who obtained samples from 100 Eldians from Wall Rose c.Hange Zoe, who obtained samples from 50 Eldians inside Wall Sina and 50 Eldians from Wall Rose d.Armin Arlert, who obtained samples from 34 Eldians from Wall Maria, 33 Eldians from Wall Rose, and 33 Eldians from Wall Sina

