Chapter14: Chromatography
Section: Chapter Questions
Problem 9P
Related questions
Question
7 please
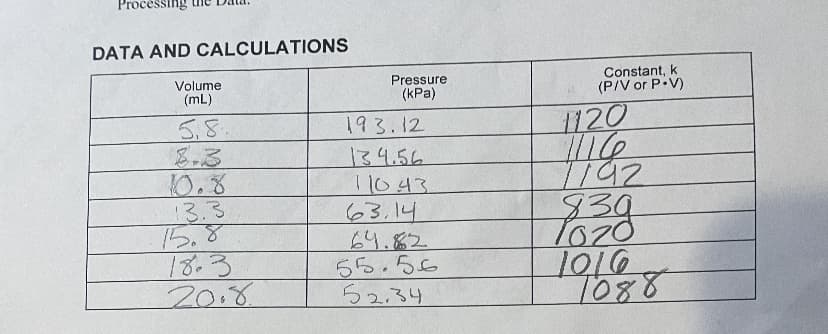
Transcribed Image Text:Processing
DATA AND CALCULATIONS
Constant, k
(P/V or P.V)
Volume
Pressure
(mL)
(kPa)
1120
5,8.
8.3
10.8
13.3
15.8
18.3
2018.
193.12
134.56
11043
63.14
64.82
55.56
ら2,34
839
1010
T088

Transcribed Image Text:7. What experimental factors are assumed to be constant in this experiment?
8. One way to determine if a relationship is inverse or direct is to find a proportionality
constant, k, from the data. If this relationship is direct, k P/V. If it is inverse, k P-V. Based
on your answer to Question 4, choose one of these formulas and calculate k for the seven
ordered pairs in your data table (divide or multiply the P and V values). Show the answers in
the third column of the Data and Calculations table.
9. How constant were the values for k you obtained in Question 8? Good data may show some
minor variation, but the values for k should be relatively constant.
10. Using P, V, and k, write an equation representing Boyle's law. Write a verbal statement that
correctly expresses Boyle's law.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



