8. Indicate whether the following statements are True or False: a. The common name for the elements in Group 1A is "alkalai metals" a. T F b. All isotopes of the same element have the same number of protons b. T F CA neutron has approximately the same mass as an electron C.T F d. lonization energy tends to decrease from right to left across a period of elements e. Atomic radius tends to decrease in a group moving from top to bottom d. T F e. T F 9. Complete the following table for the neutral element isotopes: Symbol Atomic number Mass number Number of Number of mber of Protons Neutrons Electrons 99 41 29 63 18 17
8. Indicate whether the following statements are True or False: a. The common name for the elements in Group 1A is "alkalai metals" a. T F b. All isotopes of the same element have the same number of protons b. T F CA neutron has approximately the same mass as an electron C.T F d. lonization energy tends to decrease from right to left across a period of elements e. Atomic radius tends to decrease in a group moving from top to bottom d. T F e. T F 9. Complete the following table for the neutral element isotopes: Symbol Atomic number Mass number Number of Number of mber of Protons Neutrons Electrons 99 41 29 63 18 17
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter6: The Periodic Table And Atomic Structure
Section: Chapter Questions
Problem 6.67PAE
Related questions
Question
Solve all parts otherwise I will downvote
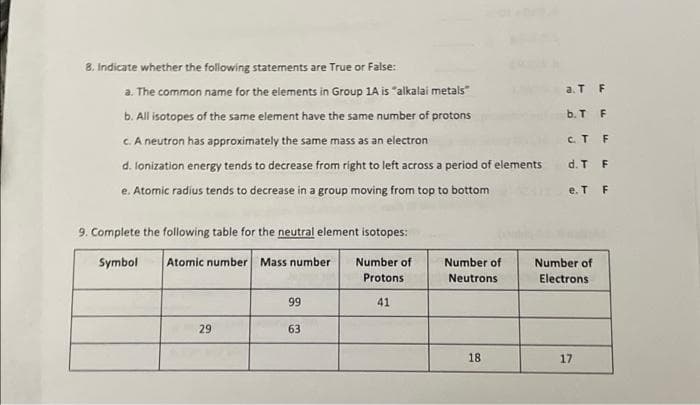
Transcribed Image Text:8. Indicate whether the following staternents are True or False:
a. The common name for the elements in Group 1A is "alkalai metals"
a. T F
b. All isotopes of the same element have the same number of protons
b. T F
C.A neutron has approximately the same mass as an electron
C.T F
d. lonization energy tends to decrease from right to left across a period of elements
d. T F
e. Atomic radius tends to decrease in a group moving from top to bottom
e. T F
9. Complete the following table for the neutral element isotopes:
Symbol
Atomic number Mass number
Number of
Number of
Number of
Protons
Neutrons
Electrons
99
41
29
63
18
17
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning