Q: For each chemical reaction listed in the first column of the table below, predict the sign (positive…
A: Since you have posted multiple questions, according to the guidelines we are providing you a…
Q: Question 8 Given: SO₂(g) + O₂(g) - 2 SO3(g) ----> O₂(g) + 2 SO₂(g) 99 kJ 49.5 kJ -99 kJ 198 kJ…
A: Pls repost question 9 with complete data..I am solving question 8 for you because it is complete..
Q: What volume in L of (2.2x10^-1) M base will be added to (1.03x10^-3) L of (5.907x10^-1) M acid so…
A:
Q: How many molecules (not moles) of NH3 are produced from 6.93×10−4 g of H2?
A:
Q: 9. An unknown homonuclear diatomic gas effuses 2.70 times faster than nasium gas (molar mass= 204.3…
A: We will solve the problem using Graham's law of diffusion.
Q: Which of the following is most likely to undergo an E2 reaction in acetone solvent? Br Br Cl C. TOH…
A:
Q: Calculate the pH of a solution containing a mixture of 0.20 M HCl and 0.20 M HClO2 (Ka (HClO2) = 1.2…
A:
Q: 3. Provide the product(s) or reagent(s) as appropriate. CI ? 1. H₂O, H₂SO4 2. H₂CrO4 1. NaOH 2.…
A: We have to give the reagents and products of the given reactions. Introduction:…
Q: Combustion of hydrocarbons such as decane (C₁0H₂2) produces carbon dioxide, a "greenhouse gas."…
A: Answer:- Firstly a balanced chemical equation is written. Then using the stoichiometry, moles of…
Q: Metal oxo reagents (like OsO4) are well-known to give syn dihydroxylation in their products (1).…
A:
Q: 5. The human eye can detect as little as 2.5*10-16 joules of light. How many photons of green…
A:
Q: Given the following data: C2H4(g) + 3 O2(g) → 2 CO2(g) + 2 H2O(l) DH = -1411.0 kJ…
A:
Q: What is the equilibrium constant value for NaC2H3O2 and how do you find its Kb?
A: Answer: Given compound is Sodium acetate which is the salt of weak acid (CH3COOH) and strong base…
Q: chemistry class, 26 liters of a 3% silver iodide solution must be mixed with a 16% solution to get a…
A: Answer: - Given 1. 26 litre of a 3% silver iodide solution. 2. X litre of a 16 % solution Solution…
Q: > > How many pi bonds are in this molecule? H₂ H- ²C=C² с A 1 H How many sigma bonds are in this…
A: We have been given two organic compounds and we have been asked to find out number of sigma and…
Q: A. What is the density of an unknown liquid if 15.00 mL of the unknown has a mass of 12.257 g? B.…
A:
Q: 1. Based on your observations: a. Which cell produces the highest cell potential? Explain. b. Which…
A: NOTE: Since you have asked multiple questions, according to the guidelines, we will provide the…
Q: What mass of water is produced from the complete combustion of 3.70×10−3 g of methane?
A: Given, mass of methane (CH4) react = 3.70 × 10-3 g mass of water (H2O) produced = ? Note: molar mass…
Q: For the reaction ABC + D ⇌ AB + CD ΔH°rxn = −50 kJ/mol and Ea(fwd) = 120 kJ/mol. Assuming a…
A: Answer:- This question is answered by using the simple concept of calculation of activation energy…
Q: Show the product of each synthetic step. Draw mechanisms. 1) NaH, then Ph 1 OH Br CO₂Et 2) DIBAH,…
A: We have to give the products of the each step with the mechanism.
Q: Why is the common ion effect and buffers just examples of LeChatlier's Principle?
A: Common ion effect It refers to decrease in the solubility of an ionic precipitate by the addition…
Q: 8. Calcium hydride, CaH₂, reacts with water to form hydrogen gas CaH₂ (s) + 2H₂O(l) → Ca(OH)₂ (aq) +…
A: Given -> CaH2(s) + 2H2O(l) ---> Ca(OH)2(aq) + 2H2(g) Volume of H2 = 256 L Pressure of H2 =…
Q: A lactic acid/lactate ion buffer solution contains 0.21 MHC₂H5O3 and 0.71 MC₂H5O₁, respectively. The…
A: pH of the buffer solution can be calculated from Henderson hasselbalch equation. For acidic buffer…
Q: 3- compound a ↓ compound b m 9 Reagents a. HCI b. HBr CH₂O. H₂SO4 d. Br₂ e. Ch 1. 9 h. i. J. k. 1.…
A: Answer : The Reagents used are Reagent : eReagent: m Reagent: q The compound b & c is given…
Q: A sample of nitrogen gas inside a sealed container with a volume of 6.0 liters and temperature of…
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: Write a balanced thermochemical equation with phase labels for the Haber process with the heat…
A:
Q: For the following reacrion sequence, what are the correct structures of products A through D? OH or…
A: Synthesis of alcohol Trimethyl silane act as protecting group for alcohol.TBAF act as deprotecting…
Q: f 5.42 g of CuNO3 is dissolved in water to make a 0.410 M solution, what is the volume of the…
A:
Q: A student sets up the following equation to convert a measurement. (The ? stands for a number the…
A: Answer: This question is based on unit conversion where Joule needs to be converted into kilojoules…
Q: why does m-nitroaniline dissolve in acid?
A: When a base reacts with an acid, it acquires a positive charge that causes the positively charged…
Q: A student sets up the following equation to convert a measurement. (The ? stands for a number the…
A: Here we are required to find the conversion factors which is required by the student to covert the…
Q: An unknown liquid has a heat of vaporization of 36.11 kJ/mole. If the normal boiling point is 89,…
A: We will use clausius clapeyron equation to solve this question.
Q: What are possible targets of anticancer therapies? Olipids enzymes Onucleic acids Oglycans
A: Numerous medications used to treat cancer use various mechanisms of action. Targeting drugs at…
Q: While the majority component of air is nitrogen (N2 ), the gas is very unreactive because of its…
A:
Q: (a) How many milliliters are contained in the dose of Children's Tylenol shown in the adjacent photo…
A: a.) We can read the value of volume from the given image. b.) Using the total volume and amount in…
Q: What mass precipitate will form if 0.300 L of 0.190 M Nal is mixed with excess Pb(ClO3)2?…
A:
Q: A chemical engineer is studying the two reactions shown in the table below. In each case, he fills a…
A:
Q: Select all of the functional groups that are present in the structure of the biomolecule below. OH…
A: The given structure is chair form of carbohydrate..
Q: Part A: A 252-mL buffer solution is 0.420 M in ammonia (NH3) and 0.303 M in ammonium chloride…
A: We would use Henderson Hasselbalch equation to calculate initial pH. Then , HBr which is a strong…
Q: 4 H₂C H3C C=0 + NaBH4 + 4 CH₂OH 38 g/mol 58 g/mol A. 51.7% B. 78.9% 4 C. 50% D. 25% E. 75% H3C OH…
A:
Q: (7 x 10-5) (6.30 × 10¹) (7.76 x 10-8) Express your answer using the correct number of significant…
A: A). After calculating , upto 3 significant figure 5.68 * 107
Q: What would be the best sequence of reactions/reagents to use for this transformation (the one that…
A: Bulky base abstracts proton from the less hindered site HBr, peroxide give anti-Markovnikoff product…
Q: 3. Provide the product(s) or reagent(s) as appropriate. ? 1. H₂O, H₂SO4 2. H₂CrO4 1. NaOH 2. H₂CRO4…
A: We have find out the products or reagents in the given reactions. Note -as per our company…
Q: 9- Give IUPAC names for the following compounds: H3C CH3 || H2C=CHCHCCH3 | CH3 CH3 CH3CH2CH=CCH2CH3
A: IUPAC nomenclature: In IUPAC nomenclature a molecule's longest chain of carbons is taken as parent…
Q: "if we could extrapolate your graph to your Y-intercept, we would, knowing the shape of our graph…
A:
Q: Place the following compounds in order of decreasing melting point. (NOTE: Please enter your answer…
A: Melting point is the temperature at which the solid melts. OR The temperature at…
Q: For each of the metabolic transformations (e) through (h), determine whether the compound on the…
A:
Q: How many moles of water are produced in this reaction?
A: Solution: Octane is a well-known source of fuel gasoline. Like other hydrocarbons on combustion, It…
Q: Pyridine (below) undergoes EAS reactions at a rate much slower than benzene, and the substitution…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: What is the structure for the final product (D), in the reaction sequence shown? 9 H3C H CH3CH₂CHOH…
A: Well, First two steps are used to prepare for the Grignard reagent. and Grignard reagents are very…
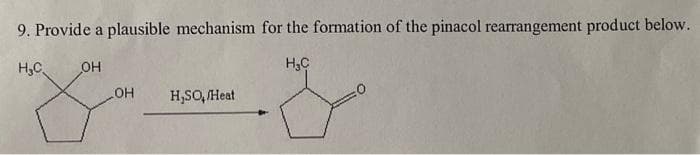
Step by step
Solved in 2 steps with 1 images

- what is the Nature of the leaving group (LG)? 5. what is the relative size of activation energy (Ea) for each reaction? 6. what is the Hammond's postulate? 7. what are the Relative thermodynamic stability of the reactive intermediates? 8. what is Influence of the solvent (if given) on the reactions and intermediates?Show the reaction mechanism for the formation of:a. RCONH2 from the reaction of RCOOR’ with NH3What type of mechanism is needed for this reaction?
- Why we need step 3 before step 4? a. Because the nitro group increases the electrophilicity at the ortho positions which is where the bromine is added. b. Because the amine group is a strong ortho, para director which is what controls the regiochemical outcome of this bromination. c. Step 4 is unessesary. The symmetry of compound 3 allows for the bromination to be regioselective and give compound 5. 5. There will be a mixture of products because there is no selectivity for a major product.What is the major product after aqueous work up?Which reacts faster in an E2 elimination, cis-or-trans1-bromo-3-tert-butylcyclohexane
- Would you expect acetate ion (CH3CO2 -) to be a better nucleophile in an SN2 reaction with an alkyl halide carried out in methanol or in dimethyl sulfoxideExplain the observed rate of reaction for the following compounds in an Sn1 reaction: (3From each pair, select the stronger nucleophile. Q.) CH3SH or CH3S-
- What is the best reagent for carrying out the reaction shown below?For SN1 Explain the order in which 1o (primary) alkyl halides reacted (fastest to slowest) and explain why. The 1o primary alkyl halides are: (see picture below) 1-chlorobutane 1-bromobutane 1-chloro-2butene benzylchloride1. Which of the following is the strongest nucleophile? Refer to the diagram below with an MeOH, MeSH, etc. 2. Which of the following is true about the stereochemistry of SN1 reaction?a.retention of configuration at the electrophilic centerb.50:50 mixture of retention and inversion of configuration at the electrophilic centerc.inversion of configuration at the electrophilic centerd. slightly more inversion than retention at the electrophilic center



