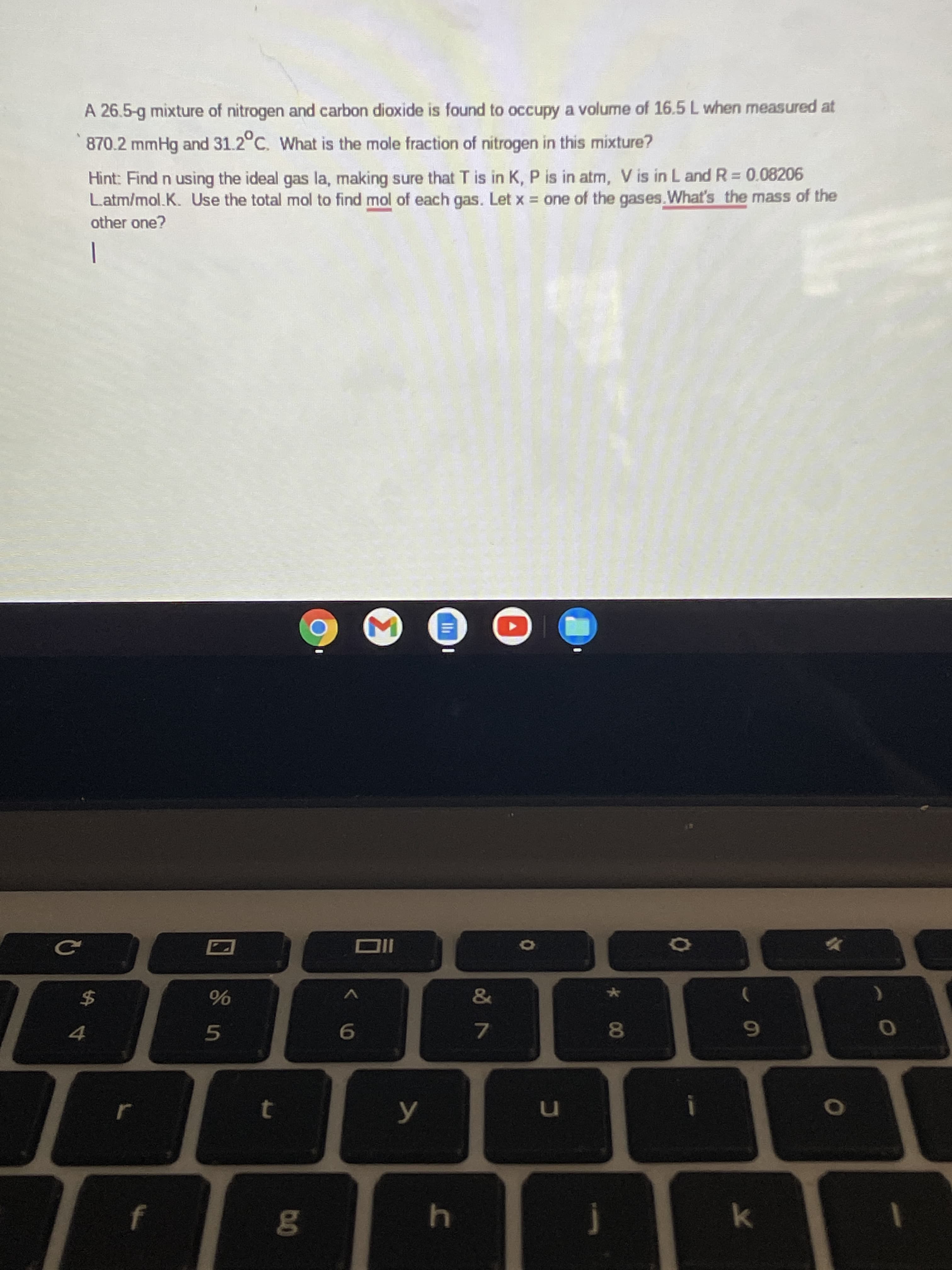
A 26.5-g mixture of nitrogen and carbon dioxide is found to occupy a volume of 16.5 L when measured at 870.2 mmHg and 31.2°C. What is the mole fraction of nitrogen in this mixture? Hint: Find n using the ideal gas la, making sure that T is in K, P is in atm, V is in L and R= 0.08206 L.atm/mol.K. Use the total mol to find mol of each gas. Let x one of the gases.What's the mass of the other one? -
A 26.5-g mixture of nitrogen and carbon dioxide is found to occupy a volume of 16.5 L when measured at 870.2 mmHg and 31.2°C. What is the mole fraction of nitrogen in this mixture? Hint: Find n using the ideal gas la, making sure that T is in K, P is in atm, V is in L and R= 0.08206 L.atm/mol.K. Use the total mol to find mol of each gas. Let x one of the gases.What's the mass of the other one? -
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter5: Gases, Liquids, And Solids
Section: Chapter Questions
Problem 5.54P: 5-54 Automobile air bags are inflated by nitrogen gas. When a significant collision occurs, an...
Related questions
Question
Transcribed Image Text:4
4
2$
8.
7.
9.
6.
IIO
|
other one?
Latm/mol.K. Use the total mol to find mol of each gas. Let x = one of the gases.What's the mass of the
Hint: Find n using the ideal gas la, making sure that T is in K, P is in atm, V is in L and R = 0.08206
%3D
870.2 mmHg and 31.2°C. What is the mole fraction of nitrogen in this mixture?
A 26.5-g mixture of nitrogen and carbon dioxide is found to occupy a volume of 16.5 L when measured at
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning