A 66.7 mL sample of 18.0 M sulfuric acid was diluted with enough water in a volumetric flask to make 5.00 x 102 mL of solution. A 25.0 ml aliquot of this solution was then further diluted to a volume of 1.50 x 102 ml. What is the molarity of the solution after the second dilution?
A 66.7 mL sample of 18.0 M sulfuric acid was diluted with enough water in a volumetric flask to make 5.00 x 102 mL of solution. A 25.0 ml aliquot of this solution was then further diluted to a volume of 1.50 x 102 ml. What is the molarity of the solution after the second dilution?
Chapter24: Introduction To Spectrochemical Methods
Section: Chapter Questions
Problem 24.18QAP
Related questions
Question
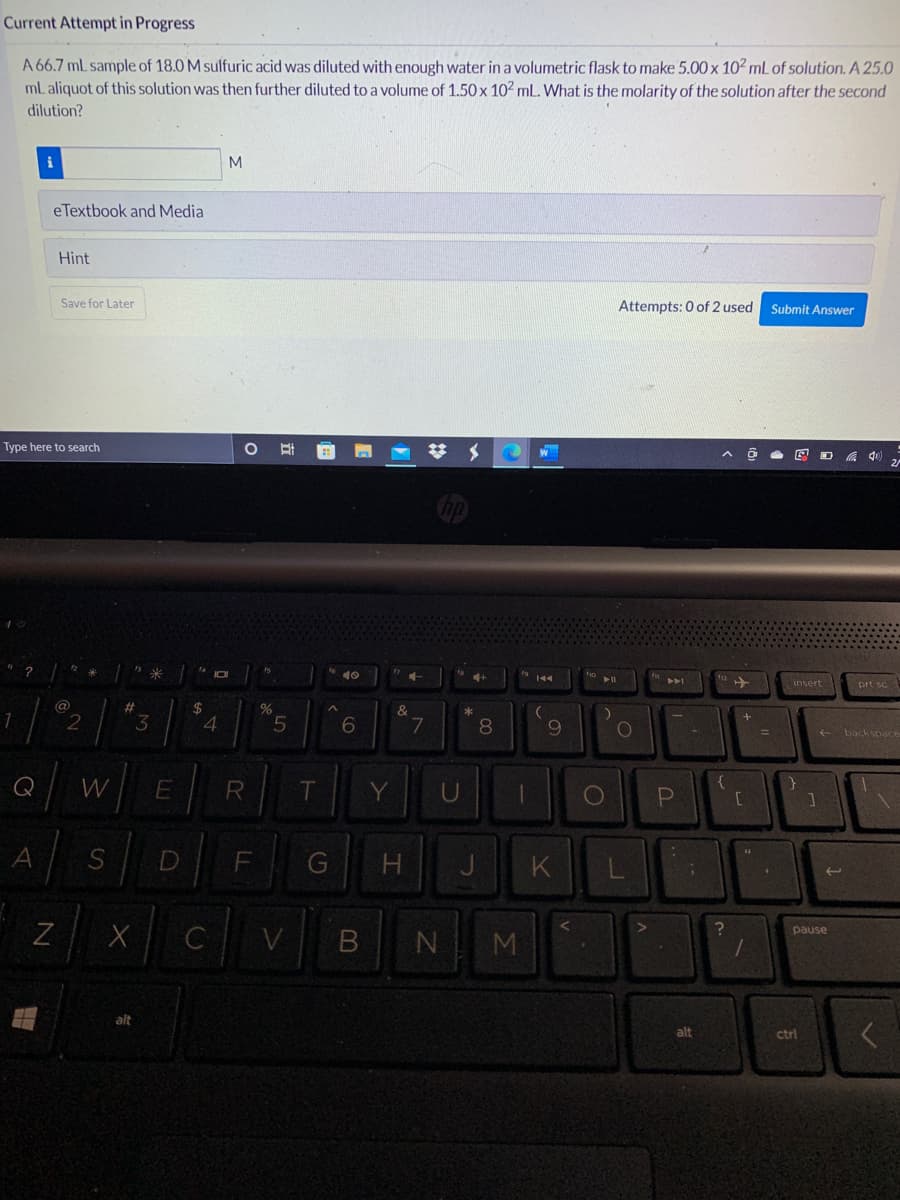
Transcribed Image Text:Current Attempt in Progress
A 66.7 mL sample of 18.0 M sulfuric acid was diluted with enough water in a volumetric flask to make 5.00 x 102 mL of solution. A 25.0
mL aliquot of this solution was then further diluted to a volume of 1.50 x 102 mL. What is the molarity of the solution after the second
dilution?
M
eTextbook and Media
Hint
Save for Later
Attempts: 0 of 2 used
Submit Answer
Type here to search
多
米
insert
prt sc
%23
%24
&
2
3
4
8
backspace
Q
W
R
Y
U
A
D
K
B N
pause
M
alt
alt
ctrl
Expert Solution
Step 1
Dilution - In dilution of the any sample the number of moles of the sample will not be changed. Change only in volume.
n1=n2
M1V1=M2V2
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

