A concept map for four types of intermolecular forces and a certain type of bond is shown. Compare the relative strength of the two forces B and C.explain how you determine this comparison but identifying the forces.
A concept map for four types of intermolecular forces and a certain type of bond is shown. Compare the relative strength of the two forces B and C.explain how you determine this comparison but identifying the forces.
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.19QAP
Related questions
Question
- A concept map for four types of intermolecular forces and a certain type of bond is shown. Compare the relative strength of the two forces B and C.explain how you determine this comparison but identifying the forces.
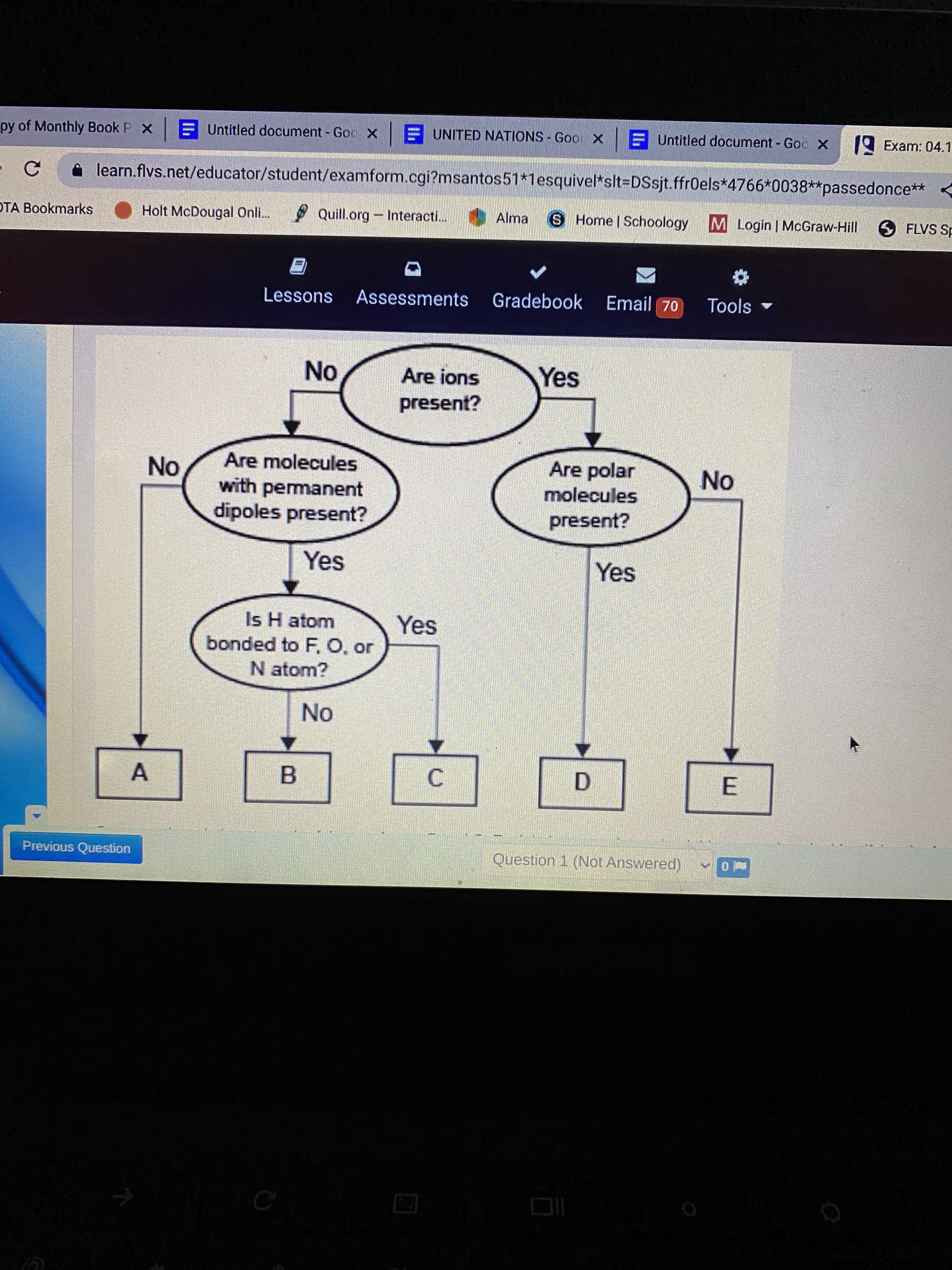
Transcribed Image Text:B
A.
py of Monthly Book P x E Untitled document - Goc X E UNITED NATIONS Goo X E Untitled document - God x
A Exam: 04.1
A learn.flvs.net/educator/student/examform.cgi?msantos51*1esquivel*slt=DSsjt.ffr0els*4766*0038**passedonce**
OTA Bookmarks
Holt McDougal Onli.
8Quill.org Interacti..
Alma
S Home Schoology
M Login | McGraw-Hill
FLVS Sp
Lessons
Assessments
Gradebook Email 70
Tools
Are ions
Yes
NO.
present?
Are molecules
Are polar
O.
with permanent
molecules
NO.
dipoles present?
present?
Yes
Yes
Is H atom
bonded to F, O, or
Yes
N atom?
O.
C.
D.
E,
Previous Question
Question 1 (Not Answered)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

