I Review | Constants | Periodi rail A What is the percent yield of the reaction? • View Available Hint(s) Hydrogen gas (a potential future fuel) can be formed by the reaction of methane with water according to the following equation: CH4(g) + H20(g) → CO(g) + 3H2(g) % In a particular reaction, 26.5 L of methane gas (measured at a pressure of 736 torr and a temperature of 25 °C) is mixed with 22.2 L of water vapor (measured at a pressure of 700 torr and a temperature of 125 °C). The reaction produces 26.4 L of hydrogen gas at STP. Submit Previous Answers X Incorrect; Try Again; 4 attempts remaining
I Review | Constants | Periodi rail A What is the percent yield of the reaction? • View Available Hint(s) Hydrogen gas (a potential future fuel) can be formed by the reaction of methane with water according to the following equation: CH4(g) + H20(g) → CO(g) + 3H2(g) % In a particular reaction, 26.5 L of methane gas (measured at a pressure of 736 torr and a temperature of 25 °C) is mixed with 22.2 L of water vapor (measured at a pressure of 700 torr and a temperature of 125 °C). The reaction produces 26.4 L of hydrogen gas at STP. Submit Previous Answers X Incorrect; Try Again; 4 attempts remaining
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter5: Gases
Section: Chapter Questions
Problem 101E: At elevated temperatures, sodium chlorate decomposes to produce sodium chloride and oxygen gas. A...
Related questions
Question
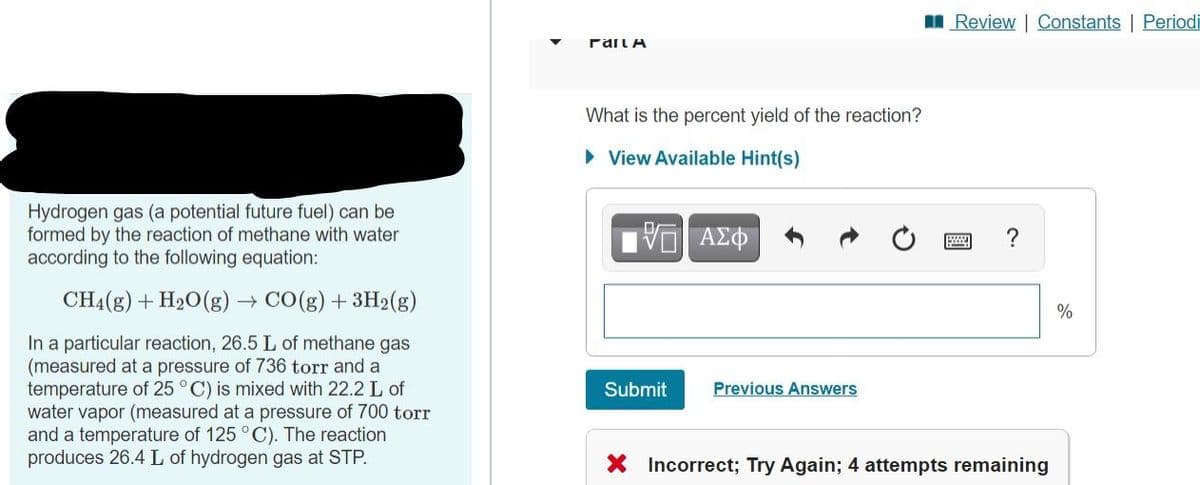
Transcribed Image Text:I Review | Constants | Periodi
rail A
What is the percent yield of the reaction?
• View Available Hint(s)
Hydrogen gas (a potential future fuel) can be
formed by the reaction of methane with water
according to the following equation:
CH4(g) + H20(g) → CO(g) + 3H2(g)
%
In a particular reaction, 26.5 L of methane gas
(measured at a pressure of 736 torr and a
temperature of 25 °C) is mixed with 22.2 L of
water vapor (measured at a pressure of 700 torr
and a temperature of 125 °C). The reaction
produces 26.4 L of hydrogen gas at STP.
Submit
Previous Answers
X Incorrect; Try Again; 4 attempts remaining
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning