Q: Determine the pH of a buffer solution containing 0.150 M of NH3 and 1.2OM of NH&CI. K of NH3 = 1.8 x…
A: Buffer :- A solution which resists the change in its pH value when small amount of strong acid or…
Q: The decomposition of nitramide in aqueous solution at 25 °C NH,NO,(aq)-N,0(g) + H,0(I) is first…
A: Given :- NH2NO2(aq) → N2O(g) + H2O(l) Reaction is first order with respect to NH2NO2 Initial…
Q: Why did breaking the P-C single bond lead to the formation of 2 species containing a free radical…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Pentalene is a most elusive molecule and has never been isolated. The pentalene dianion, however, is…
A: To know about the stability of these compounds, it is quite important to know about the number of…
Q: Consider the reaction: 2NO(g) + O2(g)= 2NO2(g) AG° = -70.6 kJ Calculate the equilibrium constant of…
A:
Q: Determine the amount of heat (in kJ) required to convert 7.1 moles of liquid methanol at -42°C into…
A:
Q: For each structure on the left, pick an identification on the right. Clear All H2N-ÇH- -CH- -OH CH2…
A:
Q: HW: Hess' Law Ericka Rivera · Apr 20 (Edited Apr 20) 20 points Find the AH of this reaction: C(s) +…
A:
Q: Draw the expanded, or complete, structural formula for the hydrocarbon represented by the…
A:
Q: An unknown compound was obtained through steam distillation. At the end of distillation, 34.2578 g…
A: Given, mass of unknown compound = 34.2578 g mass of water = 50.00 g vapour pressure of water = 703…
Q: In an esterification reaction, a carboxylic acid reacts with an excess of alcohol in acidic…
A: Given :- pentanoic acid+ 1-propanol→ ester+ H20 To draw :- Structure of ester formed in this…
Q: A certain solution has a hydrogen ion concentration of 3.30 × 10⁻⁵ ℳ. Calculate the hydroxide ion…
A:
Q: Phosphorous reacts with bromine to form phosphorous tribromide. If 35.0 grams of bromine are reacted…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: Understand the difference between DF and concentration! What is your dilution factor if you have…
A: Dilution factor is = (Total volume of sample+ diluent) ÷ Total volume of sample…
Q: d. If light with a wavelength of 690 nm strikes the photocathode, what is the maximum kinetic energy…
A:
Q: Draw the three neutral-species of fluorescein (quinonoid, lactone, zwitterion)
A: p-quinoid, lactone and zwitter ion are the three neutral species of fluorescein.
Q: Consider the hypothetical reaction: A2 A+ A The activation energy for the decomposition of A2 is 208…
A:
Q: How many grams would a 18.615 mole sample of a substance weigh if its molar mass was 399.76 grams…
A: Formula to calculate mole:- Mole = mass ÷ molar mass
Q: A. What mass of acrylonitrile can be produced? 19. Acrylonitrile (C3H3N) is the starting material…
A: Weight of C3H6 = 15.0 gm Weight of O2 = 10.0 gm Weight of NH3 = 5.00 gm
Q: What is [H3O+] of a softdrink that has a pH of 3.400?
A: Given-> pH = 3.400
Q: 4. The following have to do with rotational partition functions. a) What are the symmetry numbers o…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Consider the standard reduction potential of the following reactions: Fe2+ (aq) + 2e → → Fe(s) E° =…
A:
Q: Consider the following reaction: Mno4 Mn2+ How many water (H20) molecules mus added to the product…
A: A redox reaction is balanced by balancing the mass and charge. In an acidic medium, the charge of…
Q: REDOX TITRIMETRY I. Balancing Redox Equations Use the Half-reaction method: a) Write the…
A:
Q: Calculate the [Cl−] ,when 1 drop (0.05 mL) of 15.0 M HCl is added to 1.0 mL of solution. To…
A: Molarity of a solution can be defined as the number of moles of the solute present in 1 litre or…
Q: Which one of the following compounds will dissolve to form a molecular solution? H2SO4 CH1206 AGNO3…
A:
Q: The temperature of an object increases by 39.4 °C when it absorbs 3715 J of heat. Calculate the heat…
A: Given, Mass of the object (m) = 393 g Change in temperature (∆T) = 39.4 °C Heat absorbed (Q) = 3715…
Q: Neutral covalent molecules are extractable because: 1- Because of its negative charge 2- Because of…
A:
Q: If 50.0 mL of Thiosulfate solution are required to titrate the l2 liberated from an excess of KI by…
A: Given: Volume of S2O32- solution = 50.0 mL. Mass of KIO3 reacted = 0.300 g.
Q: a) iBr. Bri c) [BJ" (B,]" d) I :Br:
A:
Q: If 50.0 mL of Thiosulfate solution are required to titrate the l2 liberated from an excess of KI by…
A: Given: Volume of S2O32- solution = 50.0 mL. Mass of KIO3 reacted = 0.300 g. The reactions taking…
Q: What are the identidication tests used for the group III and IV elements? Give comprehensive…
A: Identification test for group III and group IV cations are needed to be discussed.
Q: Consider the hypothetical reaction: S(g) → T(g) AS° = 8.42 JK and AH° = -17.2 kJ %3D Calculate AG°…
A:
Q: (a) Discuss polydispersity and what do you meant by PDI?
A: Polymer can be defined as a large macromolecule made up ol large number of monomers.
Q: Given that an unknown atom has (4,2,-2,-1/2) set of quantum number and a neutron count of 97.…
A:
Q: Question # 1: Design a suitable synthesis plan for the preparation of 2-cyclohexylacetic acid…
A: Note: According to our guidelines we are supposed to answer only one question. Kindly repost other…
Q: Consider the titration of 25.0 mL of 0.100 M hydrochloric acid with 0.100 M NaOH. Calculate the pH…
A: Given- Volume of HCL =25.0 mL Concentration of hydrochloric acid =0.100M Concentration of NaOH=…
Q: At 10 C, the vapour pressure of a liquid substance is 6.83 x 10-2 Pa. At 30C, the vapour pressure is…
A:
Q: Calculate the concentration of an anthracene solution which produced a fluorescence intensity (I) of…
A: Given: Fluorescence intensity (I) = 815 Irradiance of beam (P0) = 1571 Length of medium (b) = 0.868…
Q: Calculate the mole fraction of sulfuric acid (H2SO4) in 8% (w/w) aqueous H2SO4 solution. Assume that…
A: w/w % concentration of sulfuric acid = 8 %Mass of aqueous solution = 100 gHere we have to…
Q: 100.0-mL sample of a saturated PbSO4 solution was evaporated until dry. If 24 mg of solid PbSO4 was…
A: Given, Volume of slution =100 mL Mass of PbSO4 = 24mg = 24 ×10-3 g Molar mass of PbSO4 = 303.26…
Q: All-cis cyclodecapentaene is a stable molecule that shows a single absorption in its 'H NMR spectrum…
A: Cyclodecapentaene is an organic compound and a conjugated 10 pi-electron cyclic system.
Q: 4. Provide an adequate route (reaction scheme) for the following reaction HO HO
A: ->First of all there occur elimination reaction and alkene formed. ->Alkene give antimorvnikov…
Q: For the gas phase decomposition of ethyl acetate, CH;COOC,H,–→CH;COOH + C,H4 the rate constant has…
A: Minimum energy needed for reactant molecules to excite and perform reaction is known as activation…
Q: 1. Answer the following questions : A. For a 0.01 molal aqueous solution of aluminum chloride,…
A: here we are required to find the ionic strength of aluminum chloride and also find the vant Hoff…
Q: Steam distillation was performed to obtain geraniol (MM = 154.25 g/mol) from dried rose petals. At…
A: According to Dalton's law of partial pressures,The total pressure of the solution,…
Q: The following spectroscopic data were obtained for compound X (molecular formula C10H140), a natural…
A:
Q: Nicotine is a diprotic acid with Ka= 1.0 x 10-6 and Ka2= 1.3 x 10-13 Calculate the pH of a 0.020 M…
A:
Q: Predict which substance has greater molar entropy. Explain. (a) CH4(g) or CCl4(l) (b) NO2(g) or…
A: Comparison of molar entropies for different compounds
Q: Concentrated HCI is added to the CH2CI2 solution of diphenylmethanol to O Help dissolve the organic…
A:
4
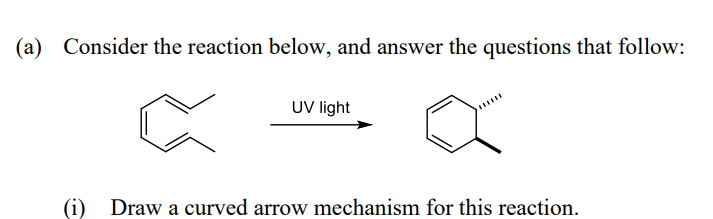
Step by step
Solved in 2 steps with 1 images

- Molecule 3 was prepared via molecules 1 and 2. Provide a complete curved arrow mechanism for the most straightforward conversion to 3. You can use HA as your acid and A- as your base.a) Provide a complete detailed mechanism (arrow pushing must be shown) b) If Benzenethiol is used in lieu of 2-Pryidemthiol, the conversion is much less effective. Explain those observations.Give the complete mechanism with arrows and the molecules below.
- Write a reasonable mechanism for the following transformation.Provide a detailed mechanism for the following (Hint:halohydrin reaction). Use arrows to show electron flow and show intermediates.Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Arrange the intermediates below in order of increasing basicity:
- Provide a detailed step-wise mechanism for the following reaction. Be sure to show all intermediates, formal charges, and show the movement of electrons with curved arrows.Provide a reasonable mechanism, including all curvy arrows and intermediates, for the following transformationConsider the following transformation below, which Prof. Ingoglia conducted during his post-doctoral research. a. Draw the product of the reaction in the box provided. b. Provide an arrow-pushing mechanism for the formation of the product you drew in part (a).

