A lab made cell consisting of an aqueous solution enclosed in a semipermeable membrane is immersed in a beaker containing a different aqueous solution. The cell membrane is permeable ONLY to water, glucose and fructose. What net movement across the cell membrane is expected if the solutions inside the cell and surrounding the cell have the concentrations described in the picture? Environment: "Cell"- 0.01 M sucrose 0.03 M sucrose 0.01 M glucose 0.01 M fructose 0.02 M glucose fructose will move from the environment into the cell O glucose will move from inside the cell out O all of these are correct fructose will move from inside the cell out O two of these are correct O three of these are correct O sucrose will move from inside the cell out
A lab made cell consisting of an aqueous solution enclosed in a semipermeable membrane is immersed in a beaker containing a different aqueous solution. The cell membrane is permeable ONLY to water, glucose and fructose. What net movement across the cell membrane is expected if the solutions inside the cell and surrounding the cell have the concentrations described in the picture? Environment: "Cell"- 0.01 M sucrose 0.03 M sucrose 0.01 M glucose 0.01 M fructose 0.02 M glucose fructose will move from the environment into the cell O glucose will move from inside the cell out O all of these are correct fructose will move from inside the cell out O two of these are correct O three of these are correct O sucrose will move from inside the cell out
Human Physiology: From Cells to Systems (MindTap Course List)
9th Edition
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Lauralee Sherwood
Chapter3: The Plasma Membrane And Membrane Potential
Section: Chapter Questions
Problem 6RE
Related questions
Question
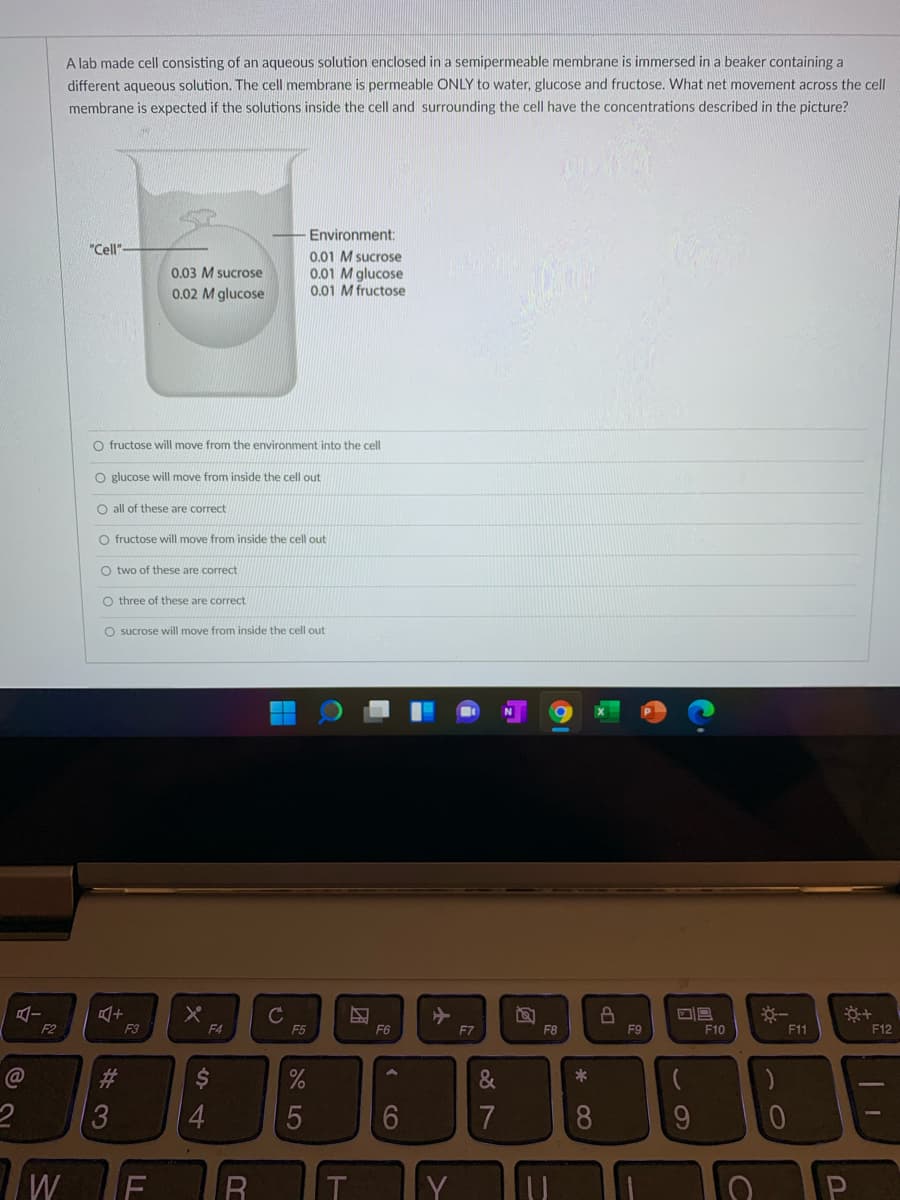
Transcribed Image Text:A lab made cell consisting of an aqueous solution enclosed in a semipermeable membrane is immersed in a beaker containing a
different aqueous solution. The cell membrane is permeable ONLY to water, glucose and fructose. What net movement across the cell
membrane is expected if the solutions inside the cell and surrounding the cell have the concentrations described in the picture?
Environment:
"Cell"-
0.01 M sucrose
0.01 M glucose
0.01 M fructose
0.03 M sucrose
0.02 M glucose
O fructose will move from the environment into the cell
O glucose will move from inside the cell out
O all of these are correct
O fructose will move from inside the cell out
O two
these are correct
O three of these are correct
O sucrose will move from inside the cell out
C
F2
F3
F4
F5
F6
F7
F8
F9
F10
F11
F12
#3
&
关
4.
6
7
8
F
5
LL
O o o oo O
శ్రీ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning