In the experimental conditions described below, how many molecules of dextrose do you have to add to the extracellular fluid in order to make it iso-osmotic relative to the intracellular fluid? Pradice g7 Intracellular fluid: • Number of water molecules = 60 • Number of Dextrose molecules = 8 • Number of Sucrose molecules = 2 Extracellular fluid: • Number of water molecules = 160 • Number of dextrose molecule = ???? • Number of Sucrose molecules = 0 Enter the number of molecules of dextrose
In the experimental conditions described below, how many molecules of dextrose do you have to add to the extracellular fluid in order to make it iso-osmotic relative to the intracellular fluid? Pradice g7 Intracellular fluid: • Number of water molecules = 60 • Number of Dextrose molecules = 8 • Number of Sucrose molecules = 2 Extracellular fluid: • Number of water molecules = 160 • Number of dextrose molecule = ???? • Number of Sucrose molecules = 0 Enter the number of molecules of dextrose
Biology Today and Tomorrow without Physiology (MindTap Course List)
5th Edition
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cecie Starr, Christine Evers, Lisa Starr
Chapter4: Energy And Metabolism
Section: Chapter Questions
Problem 2CT: Water molecules tend to diffuse in response to their own concentration gradient. How can water be...
Related questions
Question
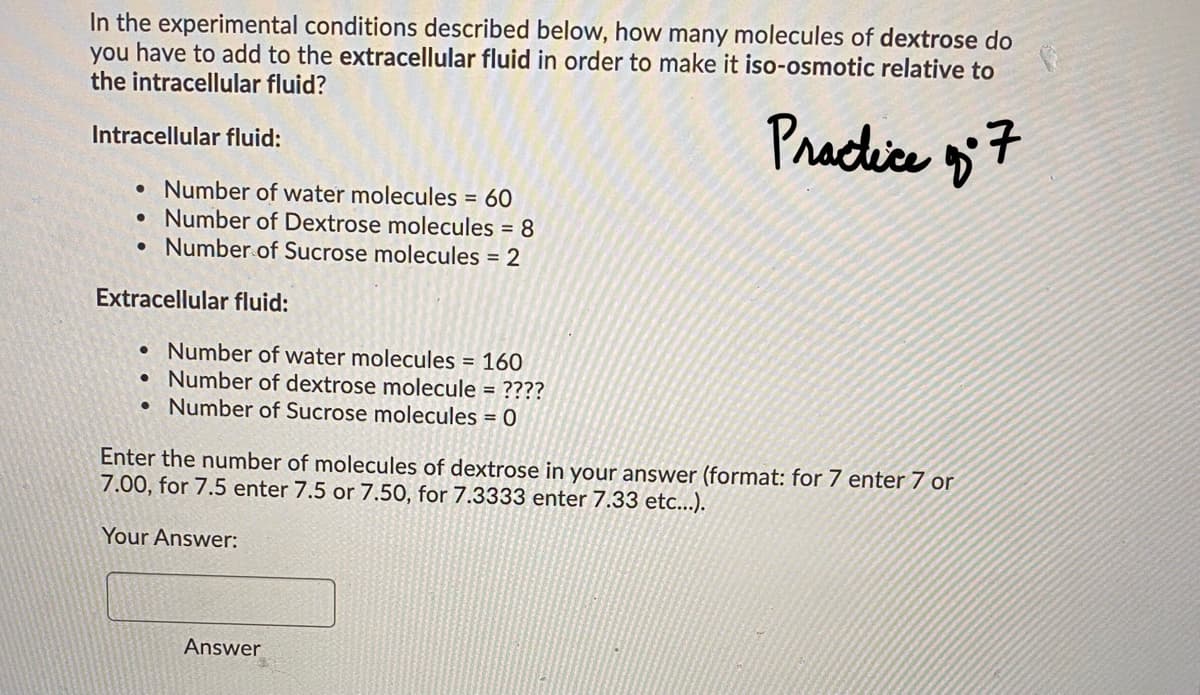
Transcribed Image Text:In the experimental conditions described below, how many molecules of dextrose do
you have to add to the extracellular fluid in order to make it iso-osmotic relative to
the intracellular fluid?
Pracice 7
Intracellular fluid:
• Number of water molecules = 60
Number of Dextrose molecules = 8
• Number of Sucrose molecules = 2
Extracellular fluid:
• Number of water molecules = 160
• Number of dextrose molecule = ????
• Number of Sucrose molecules = 0
Enter the number of molecules of dextrose in your answer (format: for 7 enter 7 or
7.00, for 7.5 enter 7.5 or 7.50, for 7.3333 enter 7.33 etc...).
Your Answer:
Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning