a) Make a table of the chemical shift, multiplicity and number of hydrogens for each 1H-NMR signal. b) Propose a structure consistent with the data and assign each proton in your structure to a signal in the NMR spectrum.
a) Make a table of the chemical shift, multiplicity and number of hydrogens for each 1H-NMR signal. b) Propose a structure consistent with the data and assign each proton in your structure to a signal in the NMR spectrum.
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter19: Nuclear Magnetic Resonance Spectroscopy
Section: Chapter Questions
Problem 19.12QAP
Related questions
Question
a) Make a table of the chemical shift, multiplicity and number of hydrogens for each 1H-NMR signal.
b) Propose a structure consistent with the data and assign each proton in your structure to a signal in the NMR spectrum.
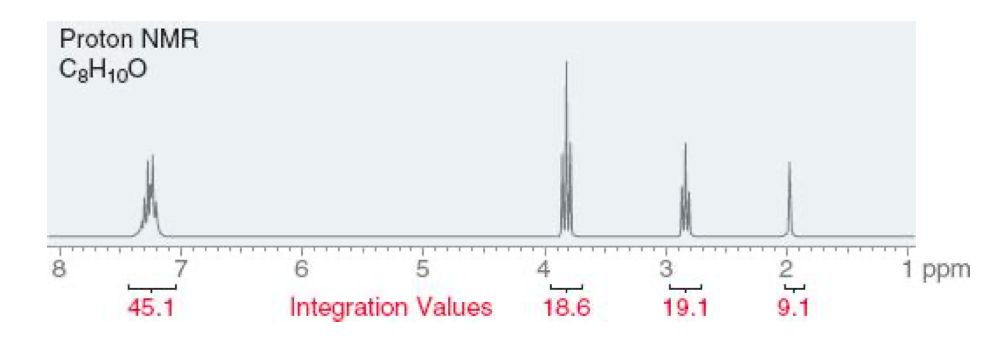
Transcribed Image Text:Proton NMR
C3H100
8
1 ppm
6.
4
3.
45.1
Integration Values
18.6
19.1
9.1
Expert Solution
Step 1: Part a
Molecule is .
Divide each signal by lowest value of integration.
9.1 corresponds to
19.1 corresponds to
18.6 corresponds to
45.1 corresponds to
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning