A sample is known to contain NaOH (MM 40.02), Na₂CO3 (MM 105.79), NaHCO, (MM 84.01) or a compatible mixture of these, together with some Inert matter. 2.000 g sample was dissolved in water making 100.0 mL solution. A 25.00 mL aliquot of this solution required 47.50 mL of 0.1800 M HCI to reach the methyl orange endpoint. A duplicate aliquot sample required 30.00 ml of the acid to reach the phenolphthalein endpoint. Identify the components of this mixed carbonates and calculate the percentage of the corresponding components in the sample.
A sample is known to contain NaOH (MM 40.02), Na₂CO3 (MM 105.79), NaHCO, (MM 84.01) or a compatible mixture of these, together with some Inert matter. 2.000 g sample was dissolved in water making 100.0 mL solution. A 25.00 mL aliquot of this solution required 47.50 mL of 0.1800 M HCI to reach the methyl orange endpoint. A duplicate aliquot sample required 30.00 ml of the acid to reach the phenolphthalein endpoint. Identify the components of this mixed carbonates and calculate the percentage of the corresponding components in the sample.
Chemical Principles in the Laboratory
11th Edition
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Chapter24: The Standardization Of A Basic Solution And The Determination Of The Molar Mass Of An Acid
Section: Chapter Questions
Problem 3ASA: A 0.3012g sample of an unknown monoprotic acid requires 24.13mL of 0.0944MNaOH for neutralization to...
Related questions
Question
5
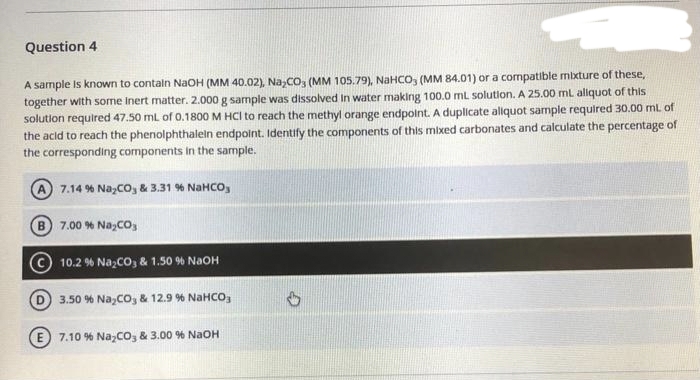
Transcribed Image Text:Question 4
A sample is known to contain NaOH (MM 40.02), Na₂CO3 (MM 105.79), NaHCO3 (MM 84.01) or a compatible mixture of these,
together with some inert matter. 2.000 g sample was dissolved in water making 100.0 mL solution. A 25.00 mL aliquot of this
solution required 47.50 mL of 0.1800 M HCI to reach the methyl orange endpoint. A duplicate aliquot sample required 30.00 ml. of
the acid to reach the phenolphthalein endpoint. Identify the components of this mixed carbonates and calculate the percentage of
the corresponding components in the sample.
A) 7.14% NayCOy & 3.31 % NaHCOy
B 7.00 % Na₂CO3
10.2 % Na₂CO3 & 1.50 % NaOH
3.50 % NayCOy & 12.9 % NaHCO,
E7.10 % Na₂CO3 & 3.00 % NaOH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole