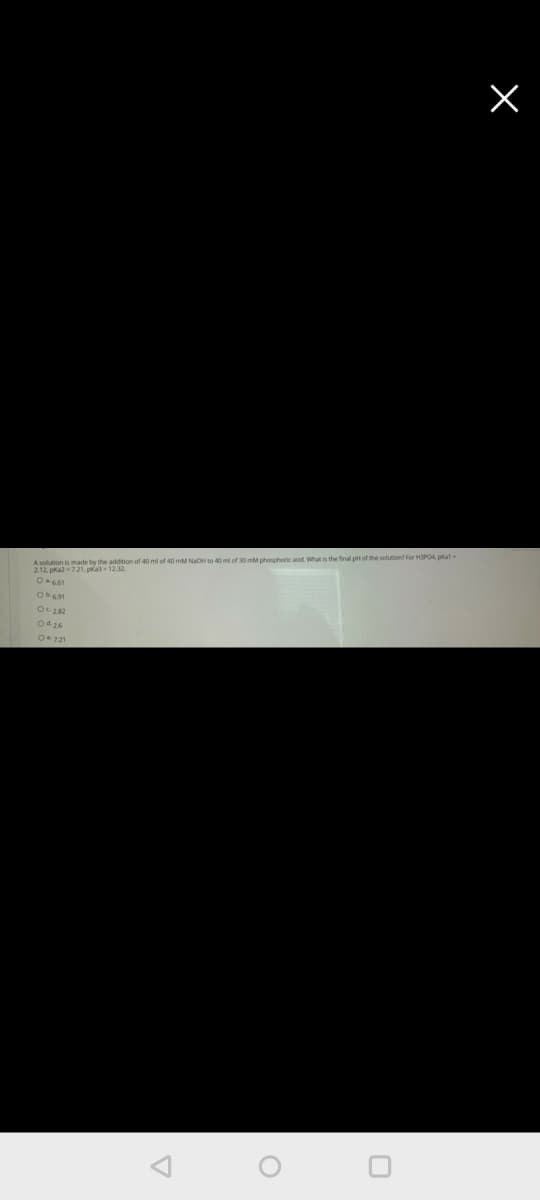
A soltion is made by the addition of 40 ml of 40 mM NaO to 40 of 30 mM phoshork acd What is the final ptof the solon for HPOA pat 2.12. pa721, pka1232 14400
Q: What is the hydronium- ion (H30*) concentration of a 2x10- MKOH solution at 25°C? O 1.0 x 10-7 M O…
A: Given that, KOH =2×10-5M at 25°C The balanced decomposition eqn is given by,…
Q: a soultion has a concentration of 6.00 and contains 1.45g of HNO3. What is the voulume of this…
A: Given Concentration = 6 mol/L mass = 1.45 g
Q: I mixed hard water or tap water or soft water 5ml + pH buffer 1ml + 3 drops of indicator + EDTA xml…
A: Hard water refers to the water sample that contains soluble salts of calcium and magnesium. It does…
Q: Calculate the number of grams of KSCN needed to make 100.0mL of 0.00200 M HSCN. Show your work:
A: Molarity
Q: How many mL of a 2.0 M NaBr solution are needed to make 200.0 mL of 0.50 M NaBr? Select one: O 50 mL…
A:
Q: Cesium chloride is used to prepare dense solutions required for isolating cellular components with a…
A: a) Cesium chloride is used to prepare dense solutions required for isolating cellular components…
Q: In standardization of HCl sample with 0.1 M Na2CO3 standard solution if we consumed 10 ml .HCl to…
A: Given: The volume of Sodium Carbonate (V1)=10 mL The volume of HCl (V2)=10 mL The molarity of Sodium…
Q: Rank the salts given in the table below from least soluble to most soluble in water. Salt #…
A: Ksp is the solubility product constant which determines solubility of a compound. The higher the…
Q: Calculate the eq per liter of 0.321g Na2CO3Na2CO3 when it is mixed in a 250ml solution. (MM Na2CO3=…
A: The normality of a solution is an important method which is used to indicate the concentration of a…
Q: alculate the Ksp of a Ca(OH)2 solution using the data below: Titrated with 0.103MHCl Initial…
A: Given Ca(OH)2 + 2HCl → CaCl2 + 2H2OMolarity of HCl = 0.103 MInitial VOlume of HCl = Vi = 9.71…
Q: Home 101 Chem 101 X (274) (Letra) A Mi Me Esta D X X х app.101edu.co Unofficial Transcript... S…
A: To find the new concentration of the solution, we have to use the dilution equation in respect to…
Q: Calculate the quantity of lime and soda required for softening one million litre of the following…
A: Given impurities are: Silica = 75 mg/lit MgCl2 = 19 mg/lit MgSO4 = 30 mg/lit CaSO4 = 68 mg/lit…
Q: A 25.0 mL sample of H3PO4 requires 50.0 mL of 1.50 M NaOH for complete neutralization. What is the…
A: Given, volume of NaOH = 50.0 ml = 0.05 L Molarity of NaOH = 1.50 M First we find the number of…
Q: What volume of HCI must be diluted to 250ml of water to obtain 0.6M solution from stock solution of…
A: Given : After preparation of solution, Volume of solution (V2)= 250 mL Concentration of HCl (M2 ) =…
Q: mL of water sample requires 2.95 mL of 0.0112 M Na25
A: According to the question, we need to calculate the oxygen concentration if the titration of 33.0 mL…
Q: Which of the following steps MUST be done prior to titration of an analyte? I. Prepare a…
A: Steps done prior titration.
Q: How many mL of 0.112M Pb(NO3)2 are needed to completely react with 25.0mL of 0.105M KI? Given:…
A: Introduction: The given chemical reaction is, PbNO32 +2 KI →PbI2 +…
Q: Q3 :Calculate the concentration of potassium ion in grams per litter after mixing 100 mL of 0.250 M…
A: Concentration of KCl = 0.250 M = 0.250 mol/L Volume of KCl = 100 mL = 0.100 L ( 1L = 1000 mL)…
Q: . If 0.045 mg of Pb2+ was observed in 1.69 kg of solution what is the ppm?
A: The question is based on the concept of solutions. We have to calculate concentration of lead in PPM…
Q: lons released Solution Conductivity of solution pH Taste solution 1 Very high 8.0 bitter ОН- 2 Very…
A: we have to say which solution is acidic.
Q: A 1.00ml aliquot of fish odl was andyrd for N using the Kieldahl methad: After chqashim, the…
A: Mili equvalent of acid=100ml×0.0503 M =5.03mili equvalent Miliequvalent of base=28.3ml×0.124M…
Q: A solution is 0.01F with respect to Mg++ ions. What concentration of hydroxide ions in formula…
A: A solution is 0.01 F with respect to Mg++ ions. Ksp of Mg(OH)2 is 1.2 * 10-11 . Here, we have to…
Q: What is the volume of HF (ideal gas at 1 atm and 25°C) required to be bubbled into 250 ml distilled…
A: pH = 2.50 -log (H)+ =2.50 H+=10-pH=10-2.50=0.00316 M
Q: Titrate 1.2035g of solid KHP (KCeHsO4) requires 23.89 mL of an unknown NaOH solution to reach the…
A:
Q: According to the table below answer the following question: Salt pair NaC KCI NaNO 3 KNO 3…
A: We have to apply equivalent conductance formula for KBr
Q: artf Homie OWLV2 Online teachin X…
A: Given, Volume of HCl solution = 37.65 mL = 0.03765 L (1 mL = 0.001 L) Mass of Na2CO3 = 2.575 g…
Q: 1. A student weighed out 0.300 grams of a monoprotic acid (HA) and added 50.0 mL of DI water to the…
A:
Q: I don't know how to answer the question to the file attached
A: According to the rules followed in significant figure calculations, the result obtained from…
Q: Doc Jill wants to determine the Ao of a weak acid she isolated called Xcitingpartic acid (HXp). She…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: 13.5 Which of the following is the best representation of a sat- urated solution? Explain your…
A: A saturated solution is a solution in which no more solute can be dissolved. But, if more amount of…
Q: 8. For a 200 mL of an aqueous solution containing 0.1 moles of KCl. If the specific conductance is…
A:
Q: Problem 1
A: The molarity of a solution is the ratio of number of moles of a substance to the volume of solution…
Q: Which of the solutes below will have i = 3? Mark all that apply. a) NH3 b) CH3OH c)…
A: The Van’t Hoff factor also denoted by ‘i’ is the ratio of the number of particles after dissociation…
Q: At 20 ml of the titrant. A) 10.23 B 11.78 12.23 D 13.78
A: Volume of HCl = 50.00 ml Molarity of HCl = 0.0275 M Molarity of NaOH = 0.1500 M Volume of NaOH = 20…
Q: To prepare 200.0 mL of 0.01 M EDTA solution, how many grams of EDTA disodium salt dihydrate…
A: Molarity is one of the concentration terms that is defined as the number of moles of solute present…
Q: While making a stock solution of NaSCN using 0.8 g of NaSCN in a 10 mL volumetric flask. 750…
A: According to the mole concept, in terms of mass, the amount of substance in moles is equal to the…
Q: Aqueo us sulfurous acid (H2S03) was made by dissolving 0.200 L of sulfur dioxide gas at 19°C and 745…
A:
Q: uves of Selected Compounds 150 140 130 120 110 100 NaNO, 90 80 70 60 50 NH CI KCI 40 Naci 30 20…
A: From the graph: It is clear that the slope of KNO3 is maximum. While the slope of other are not less…
Q: Barium sulfate is a contrast agent for X-ray scans that are most often associated with the…
A: Barium sulfate is a contrast agent for X-ray scans that are most often associated with the…
Q: Boric acid (H3BO3) is initially 3 moles/liter. Fifty milliliters of this acid solution is dikuted to…
A: Normality(N) is defined as the grams equivalent of solute present in 1000mL of the solution. N=Grams…
Q: If 20 mL of a hydrochloric acid solution (HCl) required 18 mL of a sodium hydroxide in a titration…
A: Given that, Volume of HCl = 20 mL Volume of NaOH required for neutralization of 20 mL HCl = 18 mL…
Q: 61. Concentrated HCI (36.46g/mol) has a purity of 36.5% w/w and sp.gr. 1.18; approximately how many…
A: Given that, Concentrated HCl has a purity 36.5% (w/w). The molar mass of HCl is 36.46 g/mol. The…
Q: In Step 8 (see below, as well as in the presentation), three different slimes were prepared by…
A:
Q: Solvent Isotonic Hypertonic Hypotonic What is Iodine used to detect? Describe the assay/test. What…
A: Solution- A homogeneous mixture of two or more substance is known as solution.
Q: 2- 20 ml of 0.1 M sodium hydroxide is required ---- ml of 0.1M sulphuric acid at end point 40 10 O…
A: Moles of NaOH = 2 × moles of H2SO4 Moles = molarity × volume
Q: What is the equivalence point volume of 0.140 M NAOH required to neutralize 75.0 mL of 0.350 M HC1?…
A: Concentration of NaOH=0.14M Volume=75mL Concentration of HCl=0.35M
Q: calculate the milltifres of 0-250M Naz PO that win reget with 30.0mL of O•250 M MIC12 •
A:
Step by step
Solved in 3 steps with 1 images

- 15. A 300.00 mL solution of 0.00165 M A2B5 is added to a 230.00 mL solution of 0.00380 M C2D3. What is pQsp for A2D5?Approximately 6.0mL of concentrated perchloric acid (70%) was transferred to a bottle and diluted with about 1.0L of water. A sample containing 251.5 mg of primary standard Na2B4O7.10H2O required 27.41 mL of the HClO4 solution to reach the methyl red end point. Calculate the molar concentration of the HClO4 (Fwt = 382g/mole)What is the cobalt(II) ion concentration in a solution prepared by mixing 357 mL357 mL of 0.396 M0.396 M cobalt(II) nitrate with 475 mL475 mL of 0.399 M0.399 M sodium hydroxide? The ?spKsp of cobalt(II) hydroxide is 5.92×10−155.92×10−15 .
- 2.) A 0.2185g sample of NaCl was assayed using Volhard Method with 50mL 0f 0.0998N AgNO3 and 11.9mL of 0.1350N NH4SCN. Calculate the % NaCl in the sample.5. If 0.045 mg of Pb2+ was observed in 1.69 kg of solution what is the ppm?3.) In the assay of NaHCO3, 3.0g of the solid is dissolved in 25mL water. What is the normality? How many mL of 1N H2SO4 will be required to neutralize this solution? From this volume of acid, compute the percent purity of NaHCO3.
- STANDARDIZATION OF NAOH SOLUTION A 0.4047 g sample of primary standard grade potassium hydrogen phthalate (MM = 204.23) was dissolved in distilled H2O. The solution required 22.42 mL of the NaOH solution to reach the phenolphthalein end point. a.) Calculate the number of mmoles of KHP. b.) How many moles of NaOH will react with the KHP dissolved in water? c.) Calculate the molarity of the NaOH solution.A 1.5000-g sample of cereals was analyzed for nitrogen using the Kjeldahl procedure. The receiving flask contained 69 mL of 0.02 M HCl. After the ammonia was collected, the solution was titrated with 0 M NaOH, requiring 11.72 mL to reach the methyl red endpoint. Calculate and percent protein in the sample (f=5.70). Ans. in 3 SFsYou are to make 300mL of 20mM EDTA buffer (C10H16N2O8) solution from powder. How much EDTA powder must be dissolved in water? [Atomic Weight: C = 12.011; H = 1.0079; O = 15.999; N = 14.0067]
- Explain why the conductivity of mixture A is higher than that of mixure B, , yet the pH of A is lower than the pH of B. Include relevant equations in your answer B 0.1 mol L–1 CH3COOH Conductivity (mA) 5 PH 3 A 0.1 mol L–1 HCl 260 1As solution was prepared by dissolving 5.76g of KCl.MgCl2.6H2O (277.85g/mol) in sufficient water to give 2.000L. calculate a. pMg for the solution b. pCl for the solutionA lab wishes to analyze apple juice. They would like each milliliter of titrant equal to 0.1% of malic acid. Sample aliquots will all be 10 mL. The Eqwt of malic acid is 67. What base normality should be used?

