A solution containing a mixture of metal cations was treated as follows. 1. Dilute HCl was added and a precipitate formed. The precipitate was filtered off. 2. H,S was bubbled through the acidic solution. Again, a precipitate formed and was filtered off. 3. The pH was raised to about 9 and H,S was again bubbled through the solution. No precipitate formed. 4. Finally, sodium carbonate was added and no precipitate formed. What can be said about the presence of each of these groups of cations in the original solution? Cation Group Description Present in the original solution? Ag*, Pb²+, Hg²+ form insoluble chlorides Bi3+, Cd²+, Cu²+, Hg²+, Pb2+, Sb³+, Sn²+, Snª+ form acid-insoluble sulfides Al3+, Co²+, Cr³+, Fe²+, form base-insoluble sulfides or hydroxides Fe3+, Ni2+, Mn²+, Zn²+ Ba?+, Ca?+, Mg²+, Sr²+ form insoluble carbonates
A solution containing a mixture of metal cations was treated as follows. 1. Dilute HCl was added and a precipitate formed. The precipitate was filtered off. 2. H,S was bubbled through the acidic solution. Again, a precipitate formed and was filtered off. 3. The pH was raised to about 9 and H,S was again bubbled through the solution. No precipitate formed. 4. Finally, sodium carbonate was added and no precipitate formed. What can be said about the presence of each of these groups of cations in the original solution? Cation Group Description Present in the original solution? Ag*, Pb²+, Hg²+ form insoluble chlorides Bi3+, Cd²+, Cu²+, Hg²+, Pb2+, Sb³+, Sn²+, Snª+ form acid-insoluble sulfides Al3+, Co²+, Cr³+, Fe²+, form base-insoluble sulfides or hydroxides Fe3+, Ni2+, Mn²+, Zn²+ Ba?+, Ca?+, Mg²+, Sr²+ form insoluble carbonates
Chapter17: Complexation And Precipitation Reactions And Titrations
Section: Chapter Questions
Problem 17.35QAP
Related questions
Question
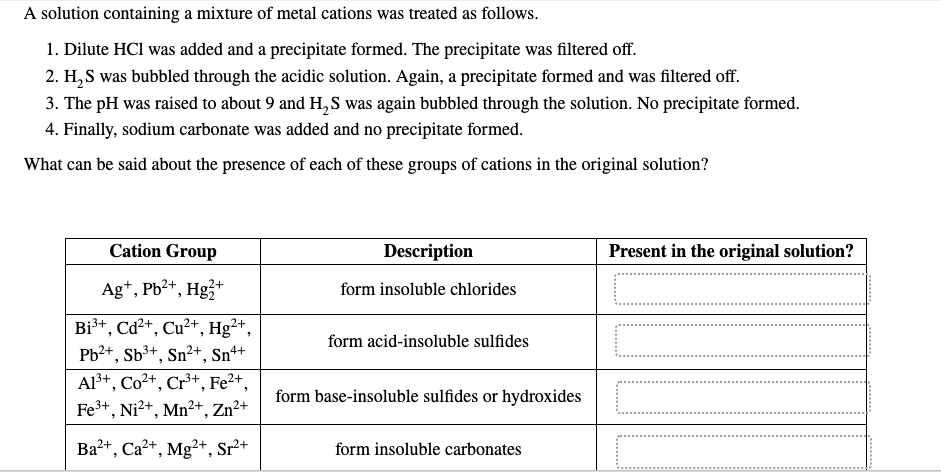
Transcribed Image Text:A solution containing a mixture of metal cations was treated as follows.
1. Dilute HCl was added and a precipitate formed. The precipitate was filtered off.
2. H,S was bubbled through the acidic solution. Again, a precipitate formed and was filtered off.
3. The pH was raised to about 9 and H,S was again bubbled through the solution. No precipitate formed.
4. Finally, sodium carbonate was added and no precipitate formed.
What can be said about the presence of each of these groups of cations in the original solution?
Cation Group
Description
Present in the original solution?
Ag+, Pb2+, Hg3+
form insoluble chlorides
Bi3+, Cd2+, Cu²+, Hg²+,
form acid-insoluble sulfides
Pb2+, Sb3+, Sn²+, Sn+
Al3+, Co2+, Cr+, Fe2+,
Fe3+, Ni?+, Mn2+, Zn²+
form base-insoluble sulfides or hydroxides
Ba2+, Ca2+, Mg²+, Sr²+
form insoluble carbonates

Transcribed Image Text:Ba?+, Ca?+, Mg²+, Sr²+
form insoluble carbonates
Lit, Na*, K*, NH
completely soluble
Answer Bank
unknown
None of these ions were present.
At least one of these ions was present.
All of these ions were present.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning