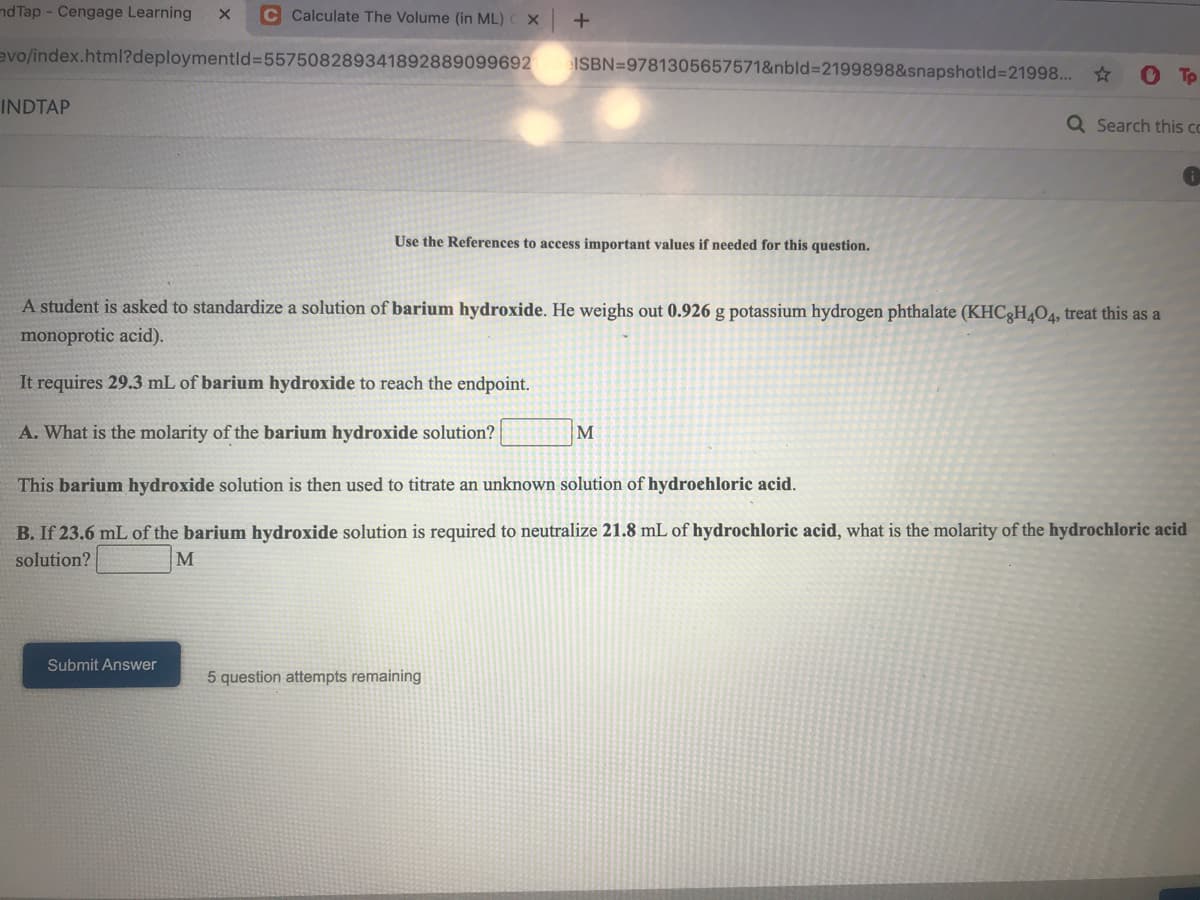
A student is asked to standardize a solution of barium hydroxide. He weighs out 0.926 g potassium hydrogen phthalate (KHC3H4O4, treat this as a monoprotic acid). It requires 29.3 mL of barium hydroxide to reach the endpoint. A. What is the molarity of the barium hydroxide solution? This barium hydroxide solution is then used to titrate an unknown solution of hydroehloric acid. B. If 23.6 mL of the barium hydroxide solution is required to neutralize 21.8 mL of hydrochloric acid, what is the molarity of the hydrochloric acid solution? M
A student is asked to standardize a solution of barium hydroxide. He weighs out 0.926 g potassium hydrogen phthalate (KHC3H4O4, treat this as a monoprotic acid). It requires 29.3 mL of barium hydroxide to reach the endpoint. A. What is the molarity of the barium hydroxide solution? This barium hydroxide solution is then used to titrate an unknown solution of hydroehloric acid. B. If 23.6 mL of the barium hydroxide solution is required to neutralize 21.8 mL of hydrochloric acid, what is the molarity of the hydrochloric acid solution? M
Chapter32: Gas Chromatography
Section: Chapter Questions
Problem 32.22QAP
Related questions
Question
Transcribed Image Text:nd Tap - Cengage Learning
C Calculate The Volume (in ML)
evo/index.html?deploymentld%3D557508289341892889099692
ISBN=9781305657571&nbld%3D2199898&snapshotld%3D21998... *
To
INDTAP
Q Search this cc
Use the References to access important values if needed for this question.
A student is asked to standardize a solution of barium hydroxide. He weighs out 0.926 g potassium hydrogen phthalate (KHC3H4O4, treat this as a
monoprotic acid).
It requires 29.3 mL of barium hydroxide to reach the endpoint.
A. What is the molarity of the barium hydroxide solution?
This barium hydroxide solution is then used to titrate an unknown solution of hydroehloric acid.
B. If 23.6 mL of the barium hydroxide solution is required to neutralize 21.8 mL of hydrochloric acid, what is the molarity of the hydrochloric acid
solution?
M
Submit Answer
5 question attempts remaining
Expert Solution
Step 1
.jpg)
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

