A student performed the fractional distillation of a cyclohexane/toluene mixture. The refractive index of the distillate was measured at 1.4301 with an ambient temperature of 22.0°C. What is the mole percentage of cyclohexane (to 3 sf)?
A student performed the fractional distillation of a cyclohexane/toluene mixture. The refractive index of the distillate was measured at 1.4301 with an ambient temperature of 22.0°C. What is the mole percentage of cyclohexane (to 3 sf)?
Biomedical Instrumentation Systems
1st Edition
ISBN:9781133478294
Author:Chatterjee
Publisher:Chatterjee
Chapter19: Biomedical Laboratory Instrumentation
Section: Chapter Questions
Problem 5P
Related questions
Question
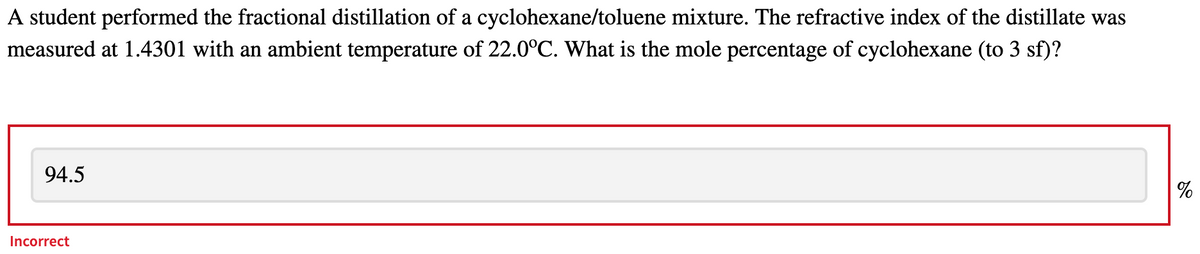
Transcribed Image Text:A student performed the fractional distillation of a cyclohexane/toluene mixture. The refractive index of the distillate was
measured at 1.4301 with an ambient temperature of 22.0°C. What is the mole percentage of cyclohexane (to 3 sf)?
94.5
Incorrect
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning


Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning