Q: Consider the electrolysis of zinc(II) bromide. Determine the product at the cathode and the product…
A: Electrochemistry is branch of chemistry in which we deal with oxidation and reduction reaction…
Q: The graph shows a heat curve for an unidentified substance discovered by the Mars rover. What is…
A: We can determine Fusion temperature ( melting or freezing point ) and vaporization temperature from…
Q: Provide a multi-step synthesis of the target structure on the right from the starting structure on…
A: Reaction 1 : Oxidation of alcohols using PCC to ketones. Reaction 2 : Aldehyde protection using…
Q: 5. (a) A galvanic cell consisting of a Cd electrode dipping into a 1.0 M CdCl2 solution and a Pt…
A: Here we have to determine the standard cell potential of the following cell in which Cd metal dipped…
Q: Which of the following is a technical challenge to using lithium metal as the anode in lithium…
A:
Q: A scientist, experimenting with liquid X, adds enough sucrose (non-volatile, non-electrolyte) to…
A: Depression in freezing point of solution = Freezing point of pure solvent – freezing point of…
Q: cell Cu(s) + Ag*(aq) → Cu*(aq) + Ag(s)
A: Given cell reaction, Cu(s) + Ag+(aq) → Cu+(aq) + Ag(s)
Q: if ) Hतर TS ववेवेटव कठ 1. 00 M नhe घृपांकिरतपका ललां् fdgo , जा which disrecton witt the equitti 2…
A: We can solve this question by using the concept of Le Chatelier principle dealing with the…
Q: Which of the following needs greater quantity of heat for 1 kg of water? a. freezing it b. Freezing…
A:
Q: What is the wavelength of light that has a frequency of 7.89 ¥ 1014 sec-1? Express the answer in…
A:
Q: 1. In the presence of an acid, triphenylcarbinol readily loses water to generate the triphenylmethyl…
A:
Q: For a given reaction ∆G°' is -27.049 V and ∆E°' is 0.2803 V. The total balanced redox reaction is…
A:
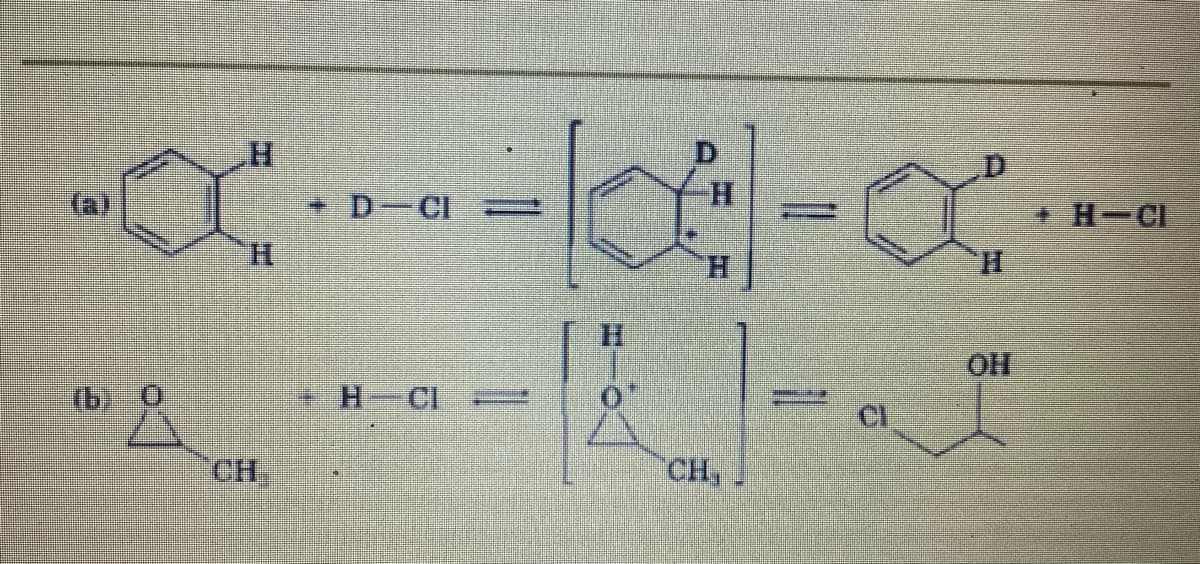
Q: D-CI H-CI H. H. H. H. (b) O H-CL CI CH,
A:
Q: WHich of the ff compouns will not form hydrogen bonding? CH3OH CH2F2 HOCH2CH2OH H2NCH2CH2CH3
A: ->Hydrogen bonds are formed when there will be such hydrogen atom which attached with…
Q: opph3 'A' CH3 Br a) BULI
A:
Q: 5. The transmittance of a solution is found to be 85.0% when measured in a cell whose path length is…
A:
Q: In the given equation: HCOOH (aq) + H2O (l) ↔ H3O+ (aq) + HCOO– (aq), the initial concentration of…
A:
Q: Calculate the percentage composition of all the atoms/elements in magnesium chlorate, Mg(CIO3)2.
A: Given :- chemical formula of Magnesium chlorate = Mg(ClO3)2 To determine :- percentage composition…
Q: 2. Draw the following structure on MolView. For each structure, obtain the following information: a.…
A:
Q: 5. Complete the following synthesis of the natural product mycophenolic acid. B. TMSCI OAc pyridine…
A: A. NaBH4 is used to reduce aldehydes to alcohols. C. DIBAL is used to convert esters to alcohols. E.…
Q: Be sure to answer all parts. For the equilibrium -8 2 H,S(g) =2 H2(g) + S2(g) K, =9.0 x 10 at 700°C…
A:
Q: Consider a galvanic cell created from the following redox reaction: 3Zn (s) + 2Au3+ (aq) → 3Zn2+…
A:
Q: Consider the following galvanic cell: Ag(s) | Ag*(3.0 x 10 4 M) || Fe3*(1.1 x 10 3 M), Fe2*(0.040 M)…
A:
Q: molecules of SO₃
A:
Q: C Experiment 10: Lewis Structures and the VSEPR Model REPORT SHEET 1. Using an appropriate set of…
A: Molecular geometry is based on hybridisation. For example all sp3 will have tetrahedral structure.…
Q: True or false The thiol group (-SH) is a better base than the hydroxide (-OH) group in elimination…
A: Since we know that H2S is more acidic than H2O...( Due to S-H bond is more weaker than O-H bond)...…
Q: Give a complete mechanism and the major product for the following reaction sequerica Please be sure…
A: Wittig reaction : Sn2 reaction between methyl bromide and TPP gives salt. Salt is treated with BuLi…
Q: A solution containing vanadium (chemical symbol V) in an unknown oxidation state was electrolyzed…
A: Given: Time =25.1 min Current passed = 1.23 A Mass of V deposited on cathode = 0.326 g Molar mass of…
Q: The hydronium ion concentration of a sample of river water is found to be 5.4 x 10-6M at 25°C. What…
A: Given, The hydronium ion concentration, H+ = 5.4 × 10-6 M at 25°C
Q: Give the IUPAC names for the following compounds. Use the abbreviations o, m, or p (no italics) for…
A:
Q: 2) Compound B (C,H30) is a carbonyl compound that give negative result on Schiff's test. CompoundB…
A: Aldehyde compounds are generally give positive result on Schiff test . Hydrolysis of ester compound…
Q: B) Benzene to N-methyl-3-phenyl propanamide Он c)
A: The synthesis of the following is:
Q: Subject: Organic Chemistry Topic: Phases of Drug Metabolism Scenario: Y.H., a 32-year-old female…
A: The above mentioned female had cronic schizophrenia. And doctor suggested risperidone which is a…
Q: A scientist, experimenting with liquid X, adds enough sucrose (non-volatile, non-electrolyte) to…
A: ★ Answer : 150.61 g of glucose is needed. ★ Solution :
Q: The rate constant k for a certain reaction is measured at two different temperatures: temperature…
A: Temperature k 106.0 °C 4.3 × 1010 192.0 °C 1.8 × 1012 Activation…
Q: Give handwritten answer. ASAP
A:
Q: 15-2 Using the Grignard reaction, carry out the following transformations. Any reagents (oganic or…
A: A. Phenyl magnesium bromide addition to cyclohexanone. B. Cyclohexyl bromide reaction with mg…
Q: 14. What is the pathlength in centimeters when the molar absorptivity for a given absorbing species…
A: Given Absorptivity = 1.32 ×103 mol-1 L cm-1 Absorbance = 0.493 Concentration= 0.000923 M l (length…
Q: In order to study hydrogen halide decomposition, a researcher fills an evacuated 2.00-L flask with…
A:
Q: 4) What is the formal concentration of NaCl (FW 58.44) when 32.0 g are dissolved in water and…
A: Formal concentration = moles of NaCl ÷ volume of solution in L
Q: yhich of the following is not a method used to protect iron objects from corrosion? O Apply a…
A: There are following methods for protection of corrosion - (1) Sacrificial protection - Here more…
Q: 3. What reagent or reagents are needed to complete the following transformations? If more than one…
A:
Q: A chemist prepares a solution of iron(III) bromide (FeBr,) by measuring out 0.13 g of FeBr, into a…
A:
Q: 3.21
A:
Q: Can someone give me the names of these two products?
A: IUPAC : Give the priority to the FG. Then start numbering the parent chain. Then give the IUPAC name…
Q: How does the Hollow Cathode Lamp work during the quantitative analysis of Iron (Fe) in Atomic…
A: Atomic absorption spectrometry (AAS) detects elements in either liquid or solid samples through the…
Q: 1. Methyl alcohol, CH,OH, has a normal boiling point of 64.7 °C and has a vapor pressure of 203 mmHg…
A:
Q: Answer the following qustion a. How many grams of aluminum bromide, AlIBr3, are present in 3.39…
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: You weigh a dry flask, and place a chunk of solid into it, then weigh it again. You then fill the…
A: The volume of the flask not occupied by the solid is needed to be find out.
Q: 1. Write the chemical reagents to complete the following chemical transformations. (These reactions…
A: Detail mechanistic pathway is given below to carry out the following conversion with appropriate…
Step by step
Solved in 2 steps with 2 images

- What volume of 1.1M sulfuric acid H2SO4 is needed to contain 12.77g?If cobalt(II) sulfate is heated too strongly, the following reaction will occur CoSO4(s) à CoO(s) + SO3(g) If you are heating a sample of CoSO4·6H2O and this reaction occurs along with dehydration, what will happen to the experimental percent water? Explain your answerCocaine (C17H21O4N) is a natural substance found in the leaves of the coca plant, which has been used for centuries as a local anesthetic and stimulant. Illegal cocaine arrives in the USA either as a pure compound or as the hydrochloride salt (C17H21O4NHCl). At 25oC, the salt is very soluble in water (2.50 kg/L), but pure cocaine is much less so (1.70 g/L). What is the maximum amount of salt (in gram) that can dissolve in 50.0 mL of water? [1 mark.] When a 25 mL aqueous solution that contains 62.50 g of the salt is treated with NaOH, the salt is converted to pure cocaine. How much water (in liters) is needed additionally to dissolve the formed pure cocaine? Round your final answer off to two decimal points. C17H21O4NHCl(aq) + NaOH(aq) → C17H21O4N(aq) + H2O(l) + NaCl(aq)
- How many mL of 0.02 M Na2S2O3 contain this number of moles?Cocaine(C17H21O4N) is a natural substance found in the leaves of the coca plant, which has been used for centuries as a local anesthetic and stimulant. Illegal cocaine arrives in the USA either as a pure compound or as the hydrochloride salt(C7H21O4NHCl). At 25 degrees celsius, the salt is very soluble in water(2.59kg/L), but pure cocaine is much less so(1.70g/L) a. What is the maximum amount of salt in grams that can dissolve in 50.0mL of water? b. When a 25mL aqueous solution that contains 62.50 g of the salt is treated with NaOH, the salt is converted to pure cocaine. How much water in liters is needed additionally to dissolve the formed pure cocaine,What is the formal concentration (expressed as mol/L 5 M) of NaCl when 32.0 g are dissolved in water and diluted to 0.500 L?

