Q: Question 5 In the electrolysis of an acid solution, oxygen can be produced by the following half-rea...
A:
Q: The attached image shows the different amounts and concentrations for an iodide clock experiment, sh...
A: General Steps to be followed: We have to get the final concentration of solutions after mixing cont...
Q: What is the pH of a 0.40 M solution of benzoic acid, C6H5COOH? (The Ka value for benzoic acid is 6.3...
A: given :- concentration of benzoic acid = 0.40 M Ka for benzoic acid = 6.3 × 10-5 To calculate :- ...
Q: rite the net ionic equation for the quilibrium that is established when ssolved in water. + H20(e) h...
A: Potassium Fluoride(KF) is a salt. It hydrolysis reaction is below, KF(aq.) + H2O(l) ---> KOH(aq.)...
Q: . Osmotic pressure is influenced primarily by an osmotic gradient between two solutions which are se...
A: When a solution is separated by semipermeable membrane from water , a pressure must be applied to so...
Q: Example: A phosphorus oxide contains 43.64% P and 56.36% O and its experimentally determined molar m...
A: Given compositions--- % P= 43.64 % % O = 56.36% Molar mass = 283.89 g mol-1 Empirical and molecular...
Q: Using molarity to find solute mass and soluu Calculate the volume in milliliters of a 1.90 mol/L iro...
A: Molarity is a concentration term for a solution. The molarity of a given solution is defined as the ...
Q: In the electrolysis of an acid solution, oxygen can be produced by the following half-reaction: 2H20...
A:
Q: For answers to calculations, use the correct number of significant figures. The percentage of sodium...
A:
Q: A 5.70-g chunk of potassium is dropped into 1.00 kg water at 24.2°C. What is the final temperature o...
A:
Q: CHEMWORK For the following statements about chemical equations, check the boxes that represent true ...
A:
Q: A solution with a mole fraction of 0.12 is composed of 0.2 mole of solute. How many moles of solvent...
A:
Q: 2. A solution may contain any of the three group I cations. A white precipitate is formed the additi...
A:
Q: Using the 13 rule and Hydrogen deficiency indicator (HDI), determine the molecular formula and propo...
A:
Q: 4. Which of the following acids, if expressed in concentration, will give the same value of molarity...
A: Malarity is the number of moles of solute present in per liter of the solution where as Normality is...
Q: A solution contains 3.86g of magnesium chloride and 250g of water. Compute for the freezing point de...
A: Note : Since you have posted multiple questions, we are entitled to answer the first only. Please re...
Q: Given the thermochemical equations A(g) → B(g) AH = 80 kJ B(g) → C(g) ΔΗ-110 kJ find the enthalpy ch...
A: Enthalpy changes for each reaction are
Q: a. An aqueous solution contains 3.35 g of iron(III) sulfate, Fe2 (SO4)3, per liter. What is the mola...
A: Given :- Mass of Fe2(SO4)3 per liter of solution = 3.35 g To calculate :- Molarity of solution an...
Q: How many Non-Polar Bonds are in the following hypothetical molecule: Electronegativity Values X: 1.3...
A: A non-polar bond has no partial positive and negative end whereas a polar bond has partial positive ...
Q: The half-equivalence point of a titration occurs half way to the equivalence point, where half of th...
A: The half-equivalence point of a titration occurs half way to the equivalence point, where half of th...
Q: A solution is made by dissolving 27.7 g of chromium(II) sulfate, CrS04, in enough water to make exac...
A: given :- mass of CrSO4 = 27.7 g volume of solution = 500 mL To calculate :- molarity...
Q: Flow Chart for the Determination of Group I Cations Ag', Pb, Hg, +6 M HCI ! centrifuge Solid AgCl, P...
A:
Q: ammonium perchlorate The pH will be less than 7. barium nitrite The pH will be approximately equal t...
A: The solutions which are acidic will have pH less than 7, more than 7 will be basic and having pH 7 w...
Q: Question 1 This following is the reaction for the synthesis of ammonia: N2 + 3 H2 + 2 NH3 At a certa...
A:
Q: The pH of an aqueous solution of 0.061 M ascorbic acid, H2C,H606 (aq), is
A: Please find your solution below : pH is the measure of extent of H+ concentration and the extent of ...
Q: The acid dissociation constant K, of hydrocyanic acid (HCN) is 6.2 x 10 -10 Calculate the pH of a 3....
A:
Q: Suppose 345. mL of dioxane and 0.23 L of toluene are mixed, and the final solution has a volume of 0...
A: Given, Volume of dioxane = 345 mL Volume of toluene = 0.23 L Final volume of the solution = 0.564...
Q: CH3 R. 10. 11. R- 12. Br 15. I B 13. 14. 16. CuIF Br
A:
Q: Part A What volume (in mL) of a 0.150 M HNO3 solution will completely react with 35.7 mL of a 0.108 ...
A: The correct answer is given below
Q: 11.35 mg KI in 110.1 mL of solution Express your answer using four significant figures. molarity = %...
A:
Q: 7.) Calculate the standard free-energy changes for the following reactions at 25°C: (a) CH4(g) + 202...
A:
Q: What number of molecules (or formula units) are present in 4.70 g of each of the following compounds...
A: We have to calculate number of molecules in 4.70g of each compounds.
Q: Flow Chart for the Determination of Group I Cations Ag', Pb, Hg, + 6 M HCI I centrifuge Solid AgCl, ...
A:
Q: Predict the FINAL (?) product (or a mixture of products) for the following synthetic transformation:...
A:
Q: The number of isomers of the Octane 1 point are twenty tow. True False 1 point B.O to PCI5 = 2. True...
A: 1) The number of isomers of the Octane are 22 . Ans : false . 2) Bond order to...
Q: Ascorbic acid (vitamin C, C6H;O6) is a diprotic acid (Ka1 = 8.0x10 and Ka2 = 1.6×10-12). What is the...
A:
Q: The solubility of rubidium nitrate in water is 150 g/L at 20 degree C. A solution is prepared at 20 ...
A: When maximum solute that can dissolve in a solution, is added to the solution, the solution is said ...
Q: Which of the following would give a positive Tollens' test? Select all that apply. 2,5-octadiene 2...
A:
Q: A chemist prepares a solution of iron(III) bromide (FeBr) by measuring out 325. g of iron(III) bromi...
A:
Q: A student ran the following reaction in the laboratory at 1180 K: 2502(0) + Oala) 2503(0) When she I...
A:
Q: Try to create a sample word problem about Hess’s Law. Include your solution following the given step...
A: The heat change in a complete chemical reaction remains same if the reaction takes place in a single...
Q: 2. What type of intermolecular force of attraction is established when sodium chloride is dissolved ...
A: The type of intermolecular force of attraction is established when sodium chloride is dissolved in p...
Q: tion 7 action solution is prepared with [S20g2] = 0.0500 M a ction is timed to the point where the r...
A: Initial concentration of [S2O82-] = 0.0500 M As it is consumed by 1.00 % So, consumed [S2O82- ] = =...
Q: Explain the major difference between the Michaelis-Menten equation and the Briggs-Haldane treatment ...
A: Micheaeli-Menten equation and Brigg-Haldane equation,both two equation describes enzyme Kinetics .
Q: Give clear handwritten Solution: Pressure of 50.0 psi is used to drive hexane through a 4.00 m leng...
A: Length of capillary = 4.00m Diameter of capillary = 7.50 um
Q: The "cartoon" shown below is an example of an rRNA cloverleaf. True False
A:
Q: Ant bonding Orbital in KOH = 4 True False O Reaction product (Cl2 + Acetylene) is polychloro .compou...
A: KOH is a ionic compound and formed by transfer of electrons.
Q: For answers to calculations, use the correct number of significant figures. The percentage of sodium...
A: To calculate mass % of NaHCO3 in given mass of sample , we would need to calculate mass of NaHCO3 in...
Q: . A solution may contain any of the three group I cations. A white precipitate is formed upon he add...
A: Analysis of group I cations .
Q: From data given below, calculate AH°, AS°, and AG° for the following reaction at 25°C. P4010 (8) + 6...
A:
Predict the major product from the following reactions and write a detailed mechanism for its formation
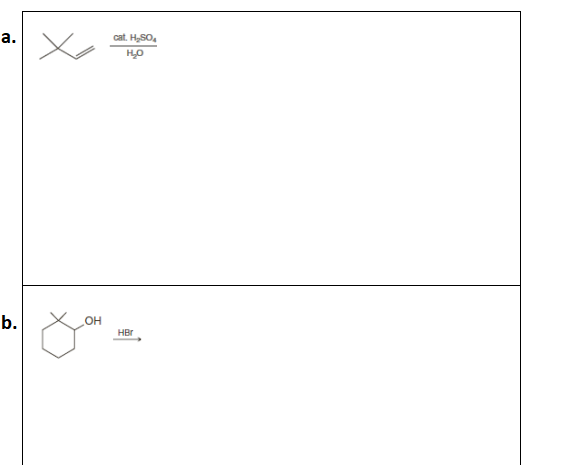
Step by step
Solved in 3 steps with 3 images

- Really hoping for solutions since I’m having a hard time with this. Pls. skip if unsure or not willing to answer the subitems (these are all connected for one item). Thanks in advanced. When the compounds analyzed by Robrob were passed on to the next test, anotherscientist, Kikoko, needed to determine the molecular weight of the active compound. They created a solution with a concentration of 20.0%w/w from the solid active compound and the solution had a density of 1.40 g/mL. From this solution, 10μL was taken and mixed with 190μL of reagent and water. It was analyzed and showed a concentration of 5x10-4 M compound.a. What is the concentration (in molarity) of the 20%w/w solution? b. Why was the concentration of the solution expressed as %w/w initially? c. What is the molecular weight of the active compound?dont provide hnadwriting solutio .....pls help and show solution plsss 46 and 47

