Abosrbance (A 280) You are interested in isolating an enzyme Ribonuclease. Inside cells ribonuclease self-associates and can exist as dimers or trimers. The active form of the enzyme is monomeric with molecular weight of ~ 14Kda. In order to isolate ribonuclease, you use size exclusion chromatography column and obtain three peaks. Indicate which peak (starting from left to right) corresponds to Monomer (M), Dimer (D) and Trimer (T) forms of ribonuclease. PEAK I: PEAK II: PEAK III: Time м Fractions
Abosrbance (A 280) You are interested in isolating an enzyme Ribonuclease. Inside cells ribonuclease self-associates and can exist as dimers or trimers. The active form of the enzyme is monomeric with molecular weight of ~ 14Kda. In order to isolate ribonuclease, you use size exclusion chromatography column and obtain three peaks. Indicate which peak (starting from left to right) corresponds to Monomer (M), Dimer (D) and Trimer (T) forms of ribonuclease. PEAK I: PEAK II: PEAK III: Time м Fractions
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter31: Completing The Protein Life Cycle: Folding, Processing, And Degradation
Section: Chapter Questions
Problem 13P
Related questions
Question
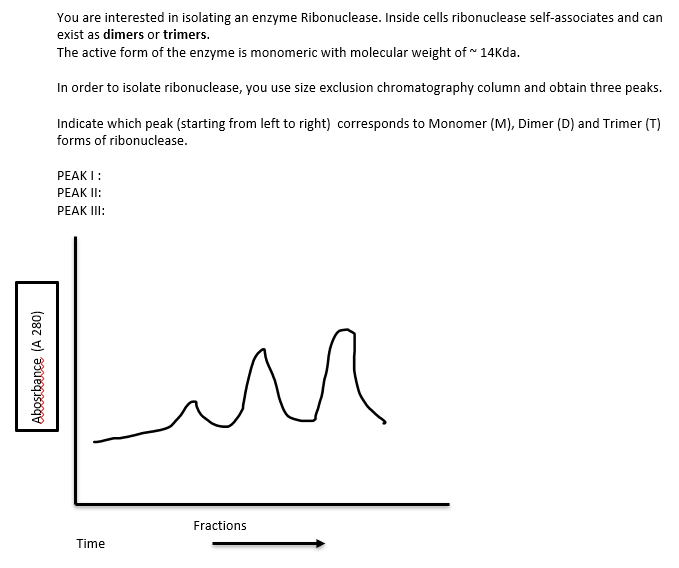
Transcribed Image Text:Abosrbance (A 280)
You are interested in isolating an enzyme Ribonuclease. Inside cells ribonuclease self-associates and can
exist as dimers or trimers.
The active form of the enzyme is monomeric with molecular weight of ~ 14Kda.
In order to isolate ribonuclease, you use size exclusion chromatography column and obtain three peaks.
Indicate which peak (starting from left to right) corresponds to Monomer (M), Dimer (D) and Trimer (T)
forms of ribonuclease.
PEAK I:
PEAK II:
PEAK III:
Time
M
м
Fractions
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning