Q: 1. Using data obtained by the explorers, try to arrange the elements together in the form of a…
A: A periodic table can only be constructed with the help of the periodicity of the chemical properties…
Q: Evaluate the energy for the two MOs generated by combining two H1s MOs. Use the equation (equation…
A:
Q: what happens when an amide reacts with water?
A:
Q: How many sigma bonds are present in the following molecule? Explain your answer.
A:
Q: A Lewis structure for the carbonate ion (CO-) is shown, but incomplete. Complete the structure by…
A:
Q: Write the complete ground-state electron configuration of B". 1s 2s 2p 3s?3p°33d104s 4p 4d 04fo…
A:
Q: ( Ni2+ ) has the following electron configuration O [Ar] 3d10 O [Ar] 3d8 O [Ar] 4s1 O [Ar] 4s1 3d9
A: In this question we have to tell the electron configuration of the Ni2+.
Q: It's also not 3.44
A: AcOH/NaOAc buffer is an acidic buffer composed of weak acid and its salt of strong base. On adding…
Q: 2) Al + HCl - AICI, + H2 Fe,(SO4); + Ba(OH),→ BaSO, +. Fe(OH), Cu CUSO,→ Al,(SO,); + Al+
A:
Q: When a sample of gas with a volume of 639.8 mL and a pressure of 0.986 atm was heated from 19.38 °C…
A:
Q: A gas is observed at the cathode and the pH also went up when a solution of KBr is electrolyzed.…
A: Given: At the cathode, gas is being formed. And the pH of the solution increased when a solution of…
Q: Question 3 Calculate the final total borate concentration (in mM) in the buffer formed by mixing…
A: Here we have to find total borate concentration in buffer solution containing 0.1650L of 110 mM of…
Q: De Broglie Hypothesis (Dual Narute of matter And Radi ohou) Ex plovn weu along with detailed…
A: Since you have multiple questions so I will solved first question for you please post separately…
Q: What type of instrument is shown in the diagram? Reference cell Po 0. Amplifier Grid Source hv…
A: The light beam in single beam instruments passes through the sample without splitting, but the beam…
Q: арply. to determine a suitable solvent system for recrystallization to monitor the progress of a…
A:
Q: What is the total number of valence electrons in the following strcuture? H || С —Н H 22 О 26 32 4 О…
A: In this question we have to tell the total number of valence electrons in the given structure.
Q: Which of the following is not a transition metal? A) Co B) Zn C) Ti D) Nb E) Tc
A: Given multiple choice question .
Q: 7. Convert 338L at 63.0 atm to its new volume at standard pressure.
A: The final volume can be calculated as follows
Q: De Broglie Hypothesis ( Dual Natuse of matter And Radi ohou) Ex plnin weu along with defailed…
A: Since, you have multiple questions so I will solved first question for you please post separately…
Q: Question 3: Ashe needs to make a total of 0.750 g of benzopinacolone. Assuming a 100% yield, how…
A: Given, mass of benzopinacolone= 0.750 g percentage yield = 100 %
Q: 22. pxygen binds to hemoglobin in which state? Plnation onh BinastoT stak= RState state b. R state…
A: Please find the below attachment.
Q: How many 3d electrons does V4+ (Vanadium) cation have in the GROUND STATE? O 3 10
A:
Q: For each of the following compounds, decide whether the compound's solubility in aqueous solution…
A:
Q: 4. Write out the steps necessary-to convert the compound on the left into the compound on the right…
A: The questions has been answered.
Q: sample of oxygen gas was collected by displacement of water in gas collection apparatus. The total…
A: Here we have to use Daltons law- Ptotal=Pwater+Poxygen It is given vapor pressure of water is 22.6…
Q: 0.750 g of Al(OH), according to the following reaction Al(OH).(s) + 3 HCI(aq) → AICI(aq) + 3 H0(aq)…
A: Here we have to calculate the mass of HCl required to reacts with 0.750 g AlCl3 according to the…
Q: When studying a 1st order reaction, you measure the concentration of the reactant at two times. at…
A: Answer: Integrated rate law for first order reaction is: k×t=2.303loga0a Here: k=rate…
Q: The decomposition of NOBr is studied manometrically (with a manometer) because the number of moles…
A: Given: Time (s) [NOBr] (M) 0.00 0.0100 2.00 0.0071 4.00 0.0055 6.00 0.0045 8.00 0.0038…
Q: 8. O2 CO2 H20 9. C,H10 O2 CO2 H20 10. C;H;OH O2 CO2 H20 + 11. C,H, O2 CO H,0
A: Count the number of atoms of each element present both side of the reaction and then, balance them.
Q: 15. Provide the product resulting from the following reaction. OsO4, NaHSO3 но OH но OH
A: Organic reactions are those in which organic reactant react to form organic products.
Q: The following information is given for antimony at 1atm: AHvap (1440.00°C) = 1.605 × 10° 1/g AHfus…
A: Answer: It is a three step process 1. Cooling of liquid antimony to its freezing point 2. Freezing…
Q: 1) LIAIH, HCI, H2O Y EN 2) H2O
A:
Q: Manipulate equation 1 to match the NO part of the overall equation. What will be the new AH value?…
A: Given equation: N2(g) + O2(g) ---------> 2NO(g) ∆H = -180.5KJ Required equation: 4NH3(g) +…
Q: Identify the disaccharides by matching the names to the structures. CH2OH CH2OH OH OH OH CH,OH CH,OH…
A:
Q: docs.google.com Which of the following is not a valid resonance structure for N3-? (V EN=N- v [一ド三司…
A: The electronic configuration of a vanadium atom is 1s2 2s2 2p6 3s2 3p6 4s2 3d3.
Q: How many meq of PO43 are there in 25.00 mL of a 2.50 M Mg3(PO4)2 solution ? The reaction is give so…
A:
Q: Label the structures to show the monosaccharide units produced by hydrolysis of the disaccharide.
A: Monosaccharides are the simplest carbohydrates that cannot be further hydrolyzed.
Q: 2-95g of NHz Jeacts, then Petial pregue N29 Temp. 298k in J-80L. container PN,=? 73H2 () 9/ mo)
A:
Q: Dinitrogen tetraoxide, a colorless gas, exists in equilibrium with nitrogen dioxide, a reddish brown…
A:
Q: C сно сно H FOH H HOH но- H HOH но —Н HO H ČH2OH CH2OH E OF сно сно H HOH HO H HO H H FOH H -OH HO H…
A:
Q: The change in Gibbs' free energy is also considered a measure of the additional work that a system…
A: Given statement is : The change in Gibbs free energy is also considered a measure of the additional…
Q: Highest S Lowest S° Answer Bank Ne(g) HF(g) F, (g) Question Source: McQuarrie, Rock, And Gallogly 4e…
A:
Q: Explain why the chemical displacement value in 1H-NMR of alkyn protons comes out between 2.5 - 3.1…
A:
Q: Determine the equilibrium constant, K. for the following process: 2A + B 2C [Aleg [Bleg = 0.0292…
A:
Q: 13.2 g of CO2 is mixed with 3.20 g of CH4 in a 4.00 L flask at a temperature of 11.2°C. What is the…
A:
Q: Determine the mass in grams of C,H10 that are required to completely react to produce 8.70 mol of…
A: Given: Moles of CO2 formed = 8.70 mol Molar mass of C4H10 = 58.12 g/mol
Q: Can someone draw the mechanism for the pericyclic mechanism and explain the stereochemistry?
A: The given substrate is N-aryl aziridine which undergoes ring opening in the presence of heat and…
Q: What thermodynamic quantity is used to determine whether a chemical reaction will proceed…
A: In the thermodynamic system we can determine which reaction is spontaneously or which reaction is…
Q: what is the number of molecules of 3Na2SO4
A: Number of molecules in 3Na2SO4 = ?
Q: Draw the major product of this reaction. Ignore inorganic byproducts. CN + H. 1. NaOH, heat 2.…
A:
Data complete
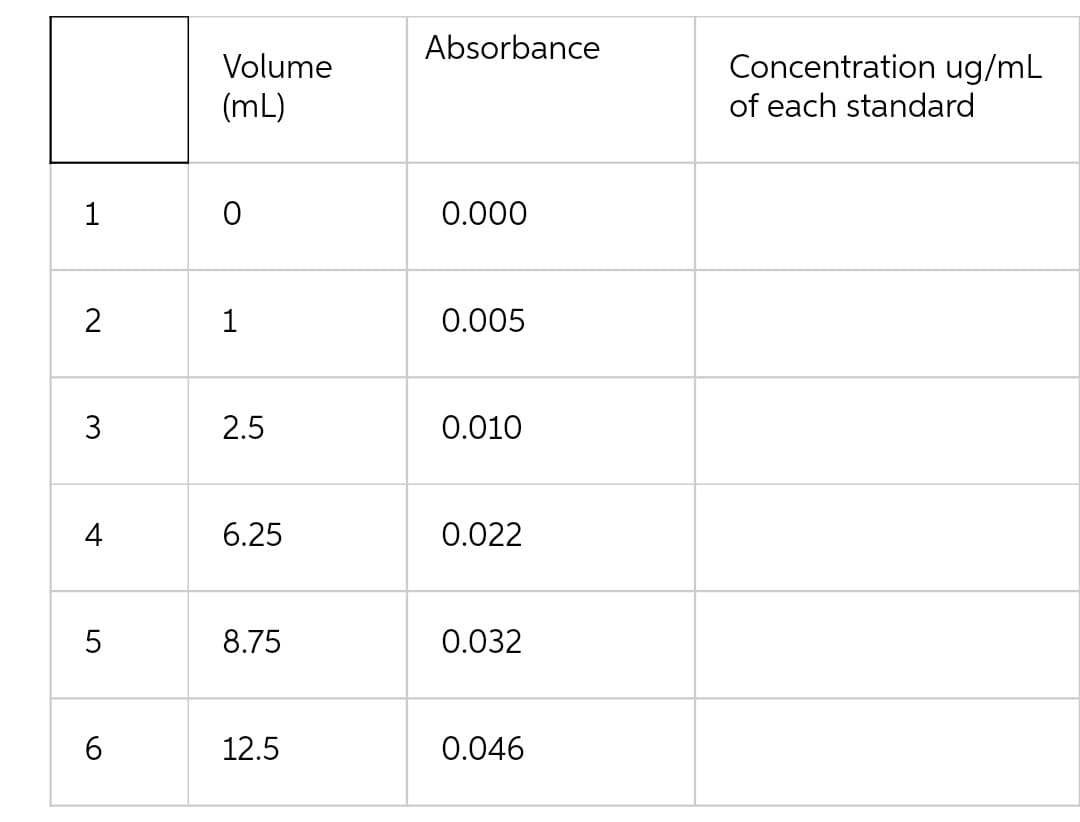
Step by step
Solved in 2 steps

- What is laser ablation and how can it he used to sample solids for ICPMS?A solution of KMnO4 with a concentration of 8.25 x 10-5 M yields an absorbance value of 0.1302 when measured in a 5.60 cm cuvette, set at a wavelength of 725 nm. Compute for the Molar Absorptivity Coefficient of the sample. Choices: a. 2.8182x10-8 c. 4.8182x10-8 b. 3.8182x10-8 d. 5.8182x10-8The absorbance values at 250nm of 5 standard solutions, and sample solution of a drug are given below: Conc. (ug/ml) A 250 nm10 0.16820. 0.32930 0.50840. 0.66050 0.846Sample. 0.661Calculate the concentration of the sample
- What percent transmittance range and what absorbance range are considered to be the optimum working ranges for spectrochemical measurements?What is the molar absorptivity of a 2.50 x 10-4 M solution if its absorbance at 550 nm is 0.500? The sample is placed in the standard 1-cm quartz cuvette. 2.00 x 10-3 M-1cm-1 5.00 x 10-4 M-1cm-1 2.00 x 103 M-1cm-1 1.25 x 10-4 M-1cm-1Plot a professional quality standard curve (absorbance vs. protein amount in µg) with an appropriate title and caption underneath the plot from the data give above. The caption should include the best-fit data.
- Application of UV- VisibleSpectroscopyConcentration [M] Absorbance Solution 1 86.29 0.64 Solution 2 51.986 0.29 …. Fig 1: [graph of the calibration curve, make sure to show: function, R2, 0 intercept]Use least square regression to determine the best straight line and slope; compare the slope from 5 & 6. Concentration (M) Absorbance (no units) 0.10 0.09 0.20 0.14 0.30 0.19 0.40 0.27 0.50 0.30 0.60 0.34 0.70 0.37
- What absorbance corresponds to 1% transmittance? To 50% transmittance?The absorbance for a dye sample of unknown concentration is measured and the corrected absorbance is 0.666. The standard curve created is found to have this linear equation: y = 0.153x + 0.043 Calculate the concentration (ug / mL) of the dye sample. 4.54 3.33 4.07 5.17 2.07Please create a caption for this table. Solution NaCl Conc. (%) Osmolality (mOsm) % transmittance Absorbance % hemolysis % crenation C distilled 0 0 0.001029 4.987584625 100 0.03354 1 0.177179111 54.61 0.001551 4.809388202 96.42720001 0.05837 2 0.297126222 91.58 0.01012 3.994819487 80.09527231 0.08444 3 0.442542222 136.4 3.849 1.414652089 28.3634704 0.134 4 0.590164444 181.9 64.8 0.188424994 3.777880643 0.2125 5 0.74752 230.4 95.64 0.019360433 0.388172513 0.3368 6 0.89644 276.3 99.56 0.001915112 0.038397585 0.5336 7 1.095648889 337.7 99.98 8.68676E-05 0.001741676 0.9834 8 1.336711111 412 100 0 0 2.1 9 1.755568889 541.1 100 0 0 7.9 10 2.674395556 824.3 100 0 0 57.83 11 4.490211111 1384 100 0 0 99.72



