Acrylonitrile and ethyl acetate have the same boiling point: 77.2°C. Which statement explains the outcome you would expect if a mixture of these two substances were distilled? * O The acrylonitrile would vaporize first and the ethyl acetate would be left behine O The ethyl acetate would vaporize first and the acrylonitrile would be left behinc O Both substances would vaporize at the same time so distillation would not be Ethyl acetate is a solid so distillation would involve allowing the acrylonitrile to evaporate.
Acrylonitrile and ethyl acetate have the same boiling point: 77.2°C. Which statement explains the outcome you would expect if a mixture of these two substances were distilled? * O The acrylonitrile would vaporize first and the ethyl acetate would be left behine O The ethyl acetate would vaporize first and the acrylonitrile would be left behinc O Both substances would vaporize at the same time so distillation would not be Ethyl acetate is a solid so distillation would involve allowing the acrylonitrile to evaporate.
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter15: Solutions
Section: Chapter Questions
Problem 17CR: Are changes in state physical or chemical changes? Explain. What type of forces must be overcome to...
Related questions
Question
100%
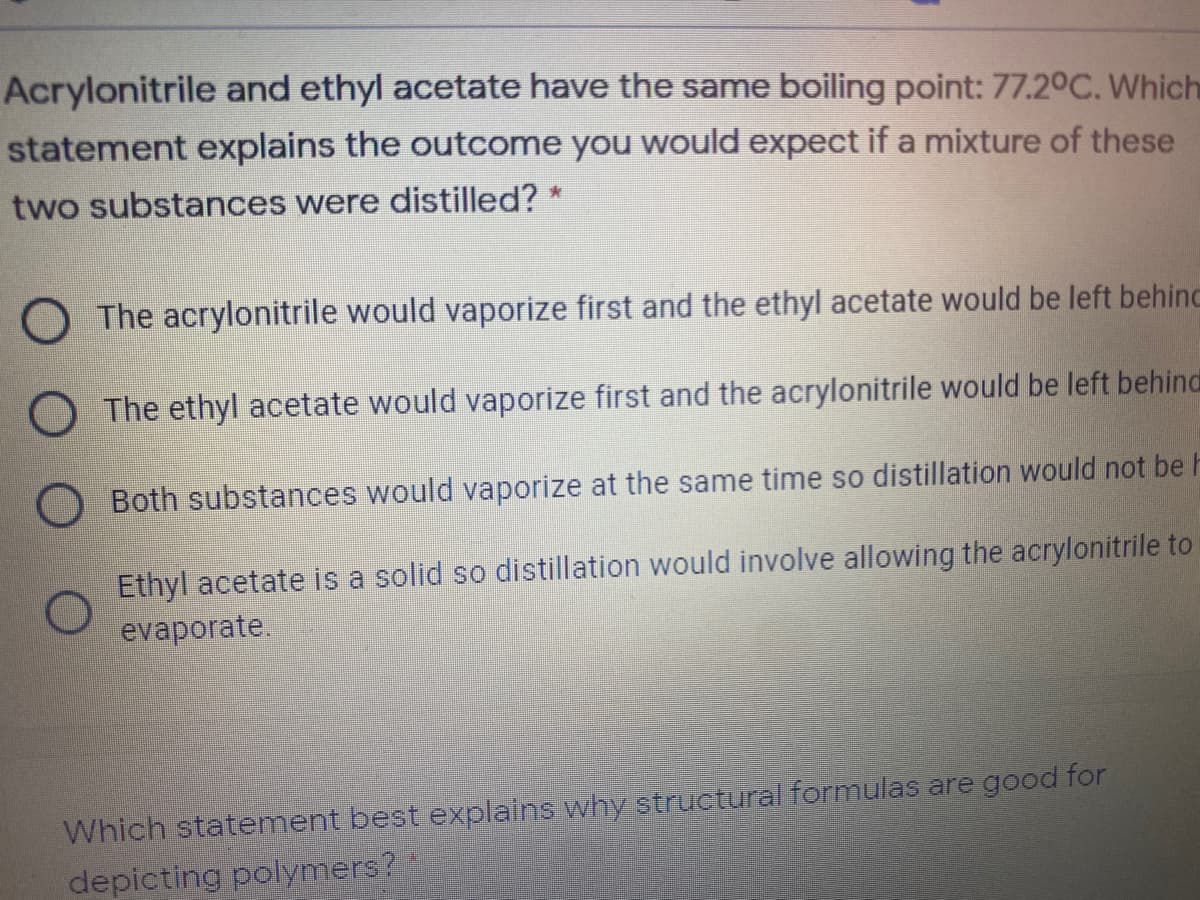
Transcribed Image Text:Acrylonitrile and ethyl acetate have the same boiling point: 77.20C. Which
statement explains the outcome you would expect if a mixture of these
two substances were distilled? *
The acrylonitrile would vaporize first and the ethyl acetate would be left behinc
O The ethyl acetate would vaporize first and the acrylonitrile would be left behind
Both substances would vaporize at the same time so distillation would not be h
Ethyl acetate is a solid so distillation would involve allowing the acrylonitrile to
evaporate.
Which statement best explains why structural formulas are good for
depicting polymers?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole