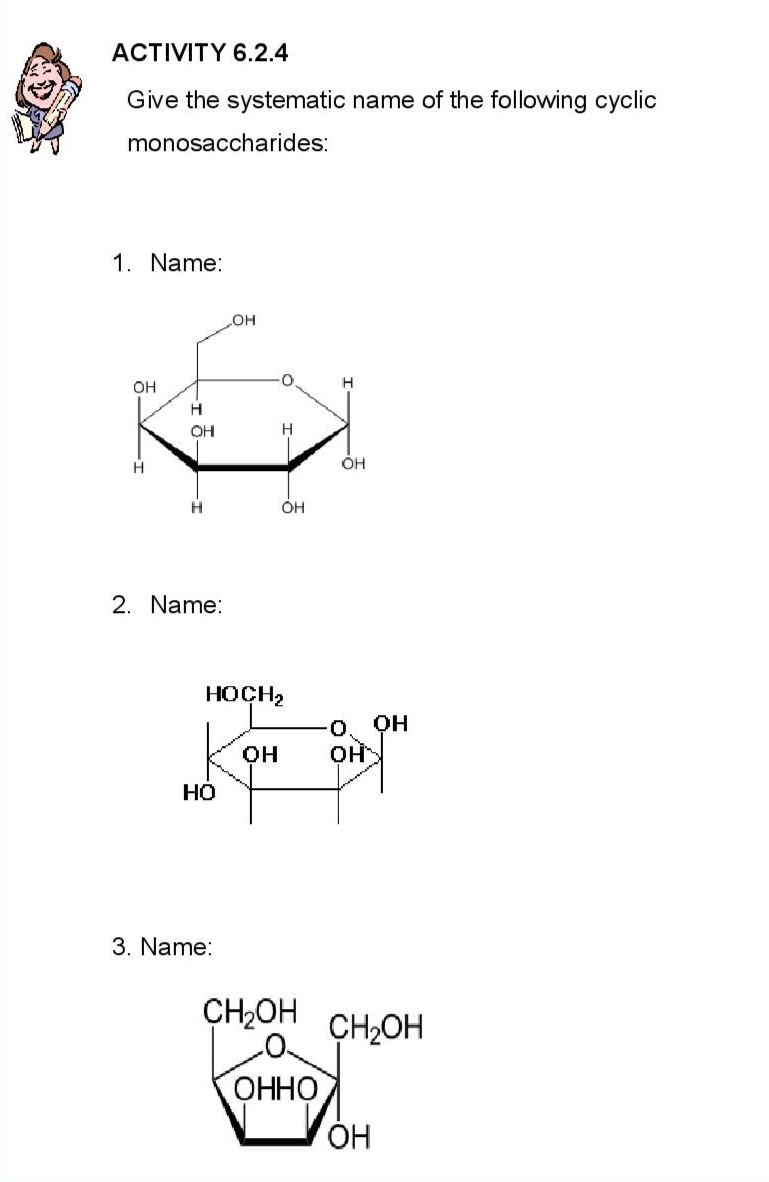
ACTIVITY 6.2.4 Give the systematic name of the following cyclic monosaccharides: 1. Name: OH Н OH Н Н 2. Name: -Т OH 3. Name: Н OH HOCH₂ OH по три види OH HO CH₂OH CH2OH OH H OH ОННО
Q: a. Is the disaccharide below a non-reducing sugar? yes or no b. The glycosidic linkage in the…
A: Carbohydrates are composed of carbon, oxygen, and hydrogen which are connected by the…
Q: What is the purpose of adding the dishwashing liquid to the DNA extraction?
A: DNA extraction is a method of separating DNA from cell membranes, proteins, and other cellular…
Q: One indication of the relative importance of various ATP-producing pathways is the Vmax of certain…
A: Pheasant pectoral muscles are clearly anaerobic, as expected from a bird highly trained for…
Q: 97) In order for a retrovirus to be infectious a. The p25 protein is cleaved by the protease enzyme…
A: Introduction: Retroviruses are a family of viruses that are grouped together based on how it is…
Q: The melting temperatures of osazones are easily determined and are characteristic for each. Listed…
A: Carbohydrates are abundant biomolecule in the nature. They are polyhydroxy aldehydes or ketones.…
Q: What is phenylketonuria? Discuss its occurrence, symptoms if any, treatments if there are, and any…
A: Phenylketonuria, also known as PKU, is an autosomal recessive disorder. Since phenylketonuria is an…
Q: Complete the table below regarding the different laboratory tests done for Carbohydrates. Only for…
A: There are different types of tests to check the presence of carbohydrates in different chemical…
Q: Which of the following is not a net product (i.e. of both phases) of the pentose phosphate shunt?…
A: Pentose phosphate pathway involves two phases , oxidative and non oxidative . It is a primary…
Q: Write the saponification reaction of the following triglyceride with NaOH Н-С-О CH3 H-C-O- `CH3…
A: Triglycerides, also known as fat molecules, are composed of long chains of fat. In the presence of…
Q: One indication of the relative importance of various ATP-producing pathways is the Vmax of certain…
A: The energy content of fat per gram is greater than that of glycogen. The tissue's ability to…
Q: How many ATP and NADH molecules would be produced during the citric acid cycle if 3 molecules of…
A: Cells are the basic unit of life. Cells derive energy from the metabolism of biomolecules such as…
Q: Which of the following contains the most lipids? A. Banana B. Champorado C. Olive oil D. Cheese
A: Different food items consist of different micro and macronutrients. Few of the macronutrients are…
Q: What aeration condition (presence or absence of O2) favors cell growth? What is the metabolic…
A: Aeration means the presence of oxygen and anaerobic condition means the absence of oxygen. The…
Q: In bacteria, single polycistronic mRNA encodes for: a single mRNA which is split into…
A: Polycistronic mRNAs are those that code for many polypeptides from a single mRNA as it involves…
Q: Compare and contrast Maillard reaction, caramelization and enzymatic browning in food.
A: A chemical reaction occurs when one or more chemicals (reactants) are converted into one or more…
Q: Below is a structure of a? monosaccharide oligosaccharide disaccharide polysaccharide
A: Carbohydrates are the most prevalent biomolecules on the planet. Carbohydrates are largely composed…
Q: How many hydrogen bonds exist between this DNA strand and its complementary strand? 5 -TGTCCCA-3'…
A: Nucleic acids are the biomolecules composed of nucleotide units. A nucleotide unit is composed of a…
Q: On a per-carbon basis, where does the largest amount of biologically available energy in…
A: There are two major types of dietary lipids: plant-derived triacylglycerol and animal-derived…
Q: In relation to the peptide sequence that is presented. -Gly - Ser – Cys – Asp – Glu – Arg – Cys –…
A: The given peptide sequence contains 8 amino acids. In a peptide, individual amino acids are joined…
Q: Two of the bypass reactions of gluconeogenesis involve: a phosphorylation of ADP using phosphate…
A: Introduction: Gluconeogenesis is the synthesis of glucose or glycogen from a non-carbohydrate…
Q: 1. The best description of how muscles move is: spinning basal bodies sliding filaments adding…
A: Muscles contract and then relax to move bodily components. Muscles can move bones, but they didn't…
Q: carbohydrate
A: Osazone test is the chemical test used to detect reducing sugars & this test allows the…
Q: for GABA transaminase, ornithine decarboxylase, and alanine
A: PLP-dependent enzymes catalyse a wide range of reactions that result in bond cleavage at the carbons…
Q: what are the dietary fats, and what are the dietary fat food and calorie densed beverage that lead…
A: Fats are made up of glycerol and fatty acids. Fats are classified into two types, saturated and…
Q: The structure of the dipeptide Gly-Asn is given by The structures of the amino acids Gly and Asn are…
A: Introduction Glycine (Gly - 3 letter code ) and Asparagine (Asn) are amino acids. Amino acids are…
Q: α-D-galactose from β-D-glucose can be differentiated using which method of analysis? A.…
A: During methylation, a methyl group gets added to every free hydroxyl (-OH) group in the sugar. This…
Q: Is it true that there is no such thing as vitamin overdose? For the fat-soluble vitamins, list down…
A: Although dying from a vitamin overdose is exceedingly rare, there were confirmed cases of death due…
Q: what is the Cartagena protocol and how do the articles of the Cartagena protocol regulate the…
A: Cartagena protocol is an international agreement on Biosafety and as a supplement to convention on…
Q: 3. Fill in the table below: Po2 percent O2 saturation of Hb (mm Hg) at pH = 7.2 at pH = 7.6 100* 40…
A: Po2 is the partial pressure of oxygen. Partial pressure is the pressure employed by a gas in a…
Q: Explain the Role of telomere in regards to cancer
A: Telomeres gradually shorten with each cell division, resulting in less protection for the…
Q: List a physiological emulsifying agent produced in the liver. What role does this substance play in…
A: Hepatocytes produce bile, which would then be altered by the cholangiocytes that line the bile…
Q: Explain the concept behind capillary electrophoresis
A: Introduction: The technique of capillary electrophoresis was first described by Jorgensen and Lukacs…
Q: Question Which of the following activities of DNA pol lis MOST important in proofreading A 5' to 3'…
A: The DNA replication in the newly added base are read by DNA pol I enzyme to check weather the added…
Q: 2. The two diagrams to the right il- lustrate plots of steady-state ki- netic studies to…
A: Phosphofructokinase adds a phosphoryl group from ATP to Fructose 6 phosphate (F6P) to yield Fructose…
Q: The Problem The three diseases in this study are borne out of errors that involve certain…
A: Introduction: Fumarase deficiency is a rare autosomal recessive metabolic disorder characterized by…
Q: Choose all that aplly that are TRUE for the lipid bilayer: Negative mark is given to incorrect…
A: A lipid bilayer is described as a very thin layer that is made up of two different layers of…
Q: do non reducing sugars have reactive anomeric carbon?
A: Introduction: Carbohydrates are polyhydroxy aldehydes or ketones with a molecular formula of CnH2On.…
Q: Compare the old (Boot's method) and new (Green) syntheses of Ibuprofen in the light of Green…
A: Introduction: Ibuprofen is a non-steroidal anti-inflammatory drug that is widely used in the…
Q: A drug was developed to inhibit the electron transport chain. How many ATP(s) would be generated by…
A: Metabolism includes biosynthesis/ reduction (an anabolic process) and oxidation (catabolic…
Q: 10 b. How many different proteins composed of 100 amino acids could possibly exist?
A: The building blocks for the synthesis of proteins are amino acids. 20 different amino acids…
Q: What are the products of the Krebs Cycle?
A: Krebs cycle is one of the most important metabolic pathway. It is also known as citric acid cycle or…
Q: Ön average, 180 liters of plasma are filtered each day. A If humans had to expend one molecule of…
A: Introduction: A mole of any substance contains as many elementary units (atoms and molecules) as the…
Q: Neurotransmitters of inhibitory signal makes the membrane potential more positive. Select one: O…
A: Hi, thank you for posting the question on Bartleby. As per the guidelines, we are authorized to…
Q: Why should starch solution be freshly prepared
A: Starch, also known as amylum, is a polymeric carbohydrate made up of several glucose units linked…
Q: The picture shown depicts what type of compound binding to an enzyme? A) A competitive inhibitor B)…
A: Regulatory enzymes show increased or decreased catalytic activity in response to specific types of…
Q: Why does saliva hydrolyze starch in the mouth but not in the stomach
A: Starch is a homo polysaccharide, produced by the plants for energy storage. Starch is composed of…
Q: B-oxidation of a molecule of palmitate (i) requires water (ii) requires NADH (iii) produces NADPH
A: "Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Which of the following statements is true regarding BoTox?
A: The question is all about the drug botox the long form is Botulinum toxin i.e Botox this drug is…
Q: (b) The results in the table below were extracted from clinical reports of pediatric age infants (3…
A: Pyruvate Dehydrogenase (PDH) complex contains 3 enzymes within it. Hence they are commonly referred…
Q: Different enzymes that catalyse the same reaction are called O A. isoenzymes O B. holoenzymes OC.…
A: Introduction: Glycolysis is the primary source of ATP production. It occurs in the cytosol of the…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- Structure activity relationship of pindololEquation for the esterification of glycerol and three ethanoic acids44. Effects of clofibrate. High blood levels of triacylglycerides are associated with heart attacks and strokes. Clofibrate, a drug that increases the activity of peroxisomes, is sometimes used to treat patients with such a condition. What is the biochemical basis for this treatment? Please Demonstrate and explain.
- RECALL Several of the enzymes of glycolysis fall into classes that we will see often in metabolism. What reaction types are catalyzed by each of the following: (a) Kinases (b) Isomerases (c) Aldolases (d) DehydrogenasesRECALL A cholesterol sample is prepared using acetyl-CoA labeled with 14C at the carboxyl group as precursor. Which carbon atoms of cholesterol are labeled?structure of cysteine at pH 8.2 and what is its charge?
- Carbohydrates Instructions: (A) Show the conversion from Fischer to Haworth projections. (B) Draw and give the systematic names of the two (2) possible Haworth structures for the following monosaccharides. 1. D- GalactoseInstructions. Given each set of information which may include common name(s) and the reaction catalyzed, you are required to identify the main class of the specific enzyme described. _____________________ Name: alkaline phosphatase Reaction: a phosphate monoester + H2O = an alcohol + phosphate _____________________ Reaction: L-threonine = D-threonine. Other information: Inverts both chiral centers, a racemase. _____________________ Name: glycine-N-acylase Reaction: acyl-COA + glycine = CoA + N-acylglycine _____________________ Name: lysine decarboxylase Reaction: L-lysine = cadaverine + CO2 _____________________ Name: methanol dehydrogenase Reaction: methanol + NAD+ = formaldehyde + NADH + H+ _____________________ Name: citryl-CoA synthetase Reaction: ATP + citrate + CoA = ADP + phosphate + (3S)-citryl-CoA _____________________ Name: D-xylulose reductase Reaction: xylitol + NAD+ = D-xylulose + NADH + H+ _____________________ Name: cellobiose phosphorylase Reaction:…Instructions. Given each set of information which may include common name(s) and the reaction catalyzed, you are required to identify the main class of the specific enzyme described. _____________________ Name: alkaline phosphatase Reaction: a phosphate monoester + H2O = an alcohol + phosphate _____________________ Reaction: L-threonine = D-threonine. Other information: Inverts both chiral centers, a racemase. _____________________ Name: glycine-N-acylase Reaction: acyl-COA + glycine = CoA + N-acylglycine _____________________ Name: lysine decarboxylase Reaction: L-lysine = cadaverine + CO2 _____________________ Name: methanol dehydrogenase Reaction: methanol + NAD+ = formaldehyde + NADH + H+
- 34.The production of glycogen within our liver and skeletal muscle cells requires what type of reaction? Dehydration Hydrolysis Ionic Dissociation. Separating Glycated Hb From Normal Hb (Integrates with Chapters 5and 6.) Human hemoglobin can react with sugars in the blood(usually glucose) to form covalent adducts. The a-amino groups ofN-terminal valine in the Hb b-subunits react with the C-1 (aldehyde)carbons of monosaccharides to form aldimine adducts, whichrearrange to form very stable ketoamine products. Quantitation ofthis “glycated hemoglobin” is important clinically, especially fordiabetic individuals. Suggest at least three methods by which glycatedHb (also referred to as HbA1c) could be separated from normal Hband quantitatedEnzyme Activity and Physiological Function, The Vmax of the enzyme glycogen phosphorylase from skeletal muscle is much greater than the Vmax of the same enzyme from liver tissue. (a) What is the physiological function of glycogen phosphorylase in skeletal muscle?





