Complete the table below regarding the different laboratory tests done for Carbohydrates. Only for the ff: •Lugol’s Test/ Iodine Test •Hydrolysis w/ Benedict’s Test •Hydrolysis w/ Iodine Test
Complete the table below regarding the different laboratory tests done for Carbohydrates. Only for the ff: •Lugol’s Test/ Iodine Test •Hydrolysis w/ Benedict’s Test •Hydrolysis w/ Iodine Test
Chapter10: Reconstitution Of Powdered Drugs
Section: Chapter Questions
Problem 7.2P
Related questions
Question
Complete the table below regarding the different laboratory tests done for Carbohydrates.
Only for the ff:
•Lugol’s Test/
Iodine Test
•Hydrolysis w/
Benedict’s Test
•Hydrolysis w/
Iodine Test
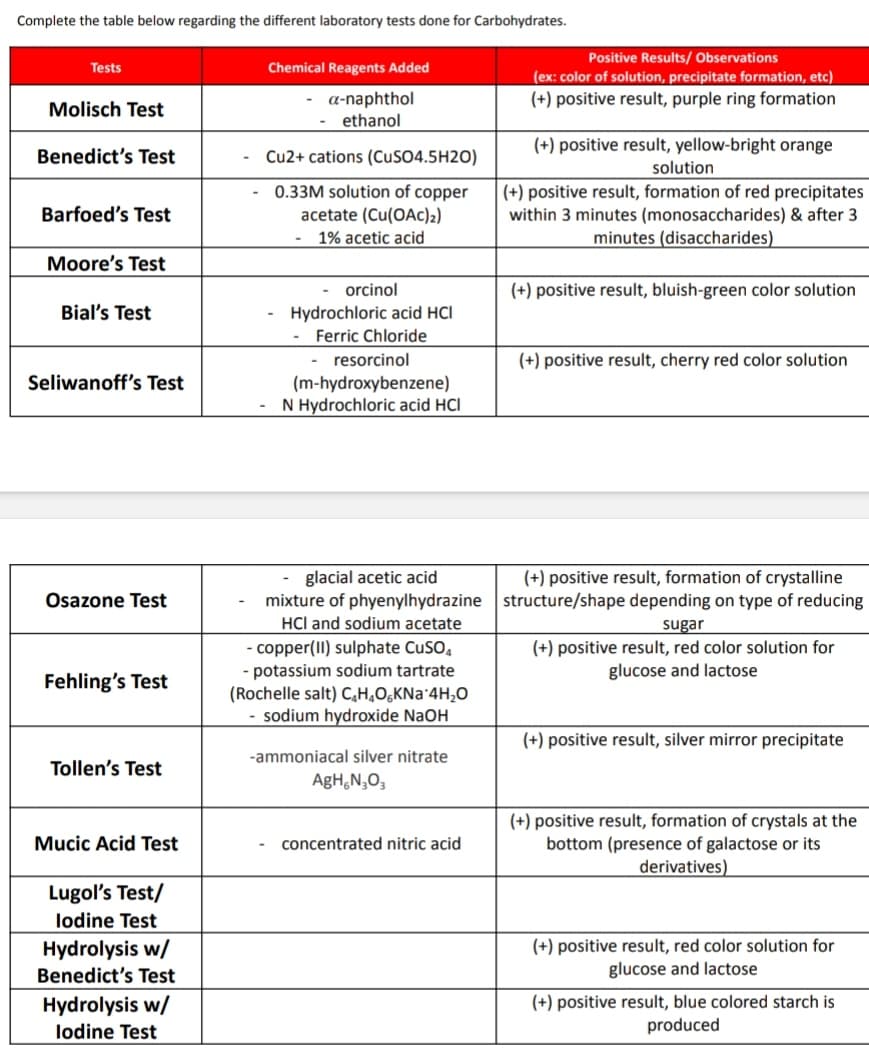
Transcribed Image Text:Complete the table below regarding the different laboratory tests done for Carbohydrates.
Tests
Chemical Reagents Added
Molisch Test
a-naphthol
ethanol
Benedict's Test
Cu2+ cations (CuSO4.5H2O)
0.33M solution of copper
Barfoed's Test
acetate (Cu(OAc)2)
1% acetic acid
Moore's Test
Bial's Test
orcinol
Hydrochloric acid HCI
Ferric Chloride
- resorcinol
(m-hydroxybenzene)
N Hydrochloric acid HCI
Seliwanoff's Test
glacial acetic acid
mixture of phyenylhydrazine
Osazone Test
HCI and sodium acetate
Fehling's Test
- copper(II) sulphate CuSO4
- potassium sodium tartrate
(Rochelle salt) C₂H₂O6KNa 4H₂O
- sodium hydroxide NaOH
Tollen's Test
-ammoniacal silver nitrate
AgH N₂O3
Mucic Acid Test
concentrated nitric acid
Lugol's Test/
lodine Test
Hydrolysis w/
Benedict's Test
Hydrolysis w/
lodine Test
Positive Results/ Observations
(ex: color of solution, precipitate formation, etc)
(+) positive result, purple ring formation
(+) positive result, yellow-bright orange
solution
(+) positive result, formation of red precipitates
within 3 minutes (monosaccharides) & after 3
minutes (disaccharides)
(+) positive result, bluish-green color solution
(+) positive result, cherry red color solution
(+) positive result, formation of crystalline
structure/shape depending on type of reducing
sugar
(+) positive result, red color solution for
glucose and lactose
(+) positive result, silver mirror precipitate
(+) positive result, formation of crystals at the
bottom (presence of galactose or its
derivatives)
(+) positive result, red color solution for
glucose and lactose
(+) positive result, blue colored starch is
produced
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Recommended textbooks for you


Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage



Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage
