Additional Exercise 19.111 An Ag/AgCl electrode dipping into 1.00 M HCI has a standard reduction potential of +0.2223 v. The half-reaction is AgC(s) + e= Ag(s) +CI(aq) A second Ag/AgCl electrode is dipped into a solution containing Cl" at an unknown concentration. The cell generates a potential of 0.0522 V, with the electrode in the solution of unknown concentration having a negative charge. What is the molar concentration of CI" in the unknown solution? [Cr] = the tolerance is +/-2% Click if you would like to Show Work for this question: Open Show Work
Additional Exercise 19.111 An Ag/AgCl electrode dipping into 1.00 M HCI has a standard reduction potential of +0.2223 v. The half-reaction is AgC(s) + e= Ag(s) +CI(aq) A second Ag/AgCl electrode is dipped into a solution containing Cl" at an unknown concentration. The cell generates a potential of 0.0522 V, with the electrode in the solution of unknown concentration having a negative charge. What is the molar concentration of CI" in the unknown solution? [Cr] = the tolerance is +/-2% Click if you would like to Show Work for this question: Open Show Work
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 66QAP: Consider a voltaic cell in which the following reaction takes place in basic medium at 25°C....
Related questions
Question
![Additional Exercise 19.111
An Ag/AgCl electrode dipping into 1.00 M HCI has a standard reduction potential of +0.2223 V.
The half-reaction is
AgCI(s) + e
Ag(s) +Cl"(aq)
A second Ag/AgCl electrode is dipped into a solution containing Cl" at an unknown concentration.
The cell generates a potential of 0.0522 V, with the electrode in the solution of unknown concentration having a negative charge.
What is the molar concentration of CI in the unknown solution?
[Cr] =
%D
the tolerance is +/-2%
Click if you would like to Show Work for this question: Open Show Work](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Fc002e5bb-f7cd-47b2-b984-337031213de7%2F99b68dda-af9e-485d-8083-e40e076070ec%2F2x63yws_processed.png&w=3840&q=75)
Transcribed Image Text:Additional Exercise 19.111
An Ag/AgCl electrode dipping into 1.00 M HCI has a standard reduction potential of +0.2223 V.
The half-reaction is
AgCI(s) + e
Ag(s) +Cl"(aq)
A second Ag/AgCl electrode is dipped into a solution containing Cl" at an unknown concentration.
The cell generates a potential of 0.0522 V, with the electrode in the solution of unknown concentration having a negative charge.
What is the molar concentration of CI in the unknown solution?
[Cr] =
%D
the tolerance is +/-2%
Click if you would like to Show Work for this question: Open Show Work
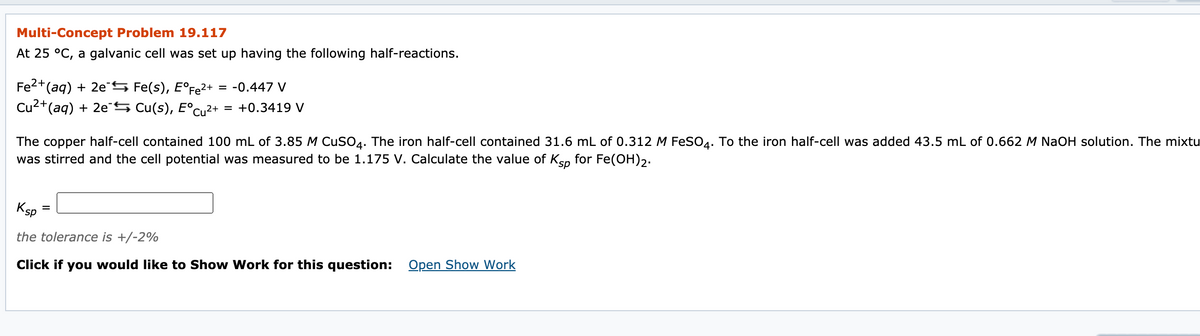
Transcribed Image Text:Multi-Concept Problem 19.117
At 25 °C, a galvanic cell was set up having the following half-reactions.
Fe2+(aq) + 2eS Fe(s), E°Fe2+
Cu2+(aq) + 2e S Cu(s), E°cu2+
= -0.447 V
= +0.3419 V
The copper half-cell contained 100 mL of 3.85 M CuSO4. The iron half-cell contained 31.6 mL of 0.312 M FeSO4. To the iron half-cell was added 43.5 mL of 0.662 M NaOH solution. The mixtu
was stirred and the cell potential was measured to be 1.175 V. Calculate the value of Ksp for Fe(OH),.
Ksp
the tolerance is +/-2%
Click if you would like to Show Work for this question: Open Show Work
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning