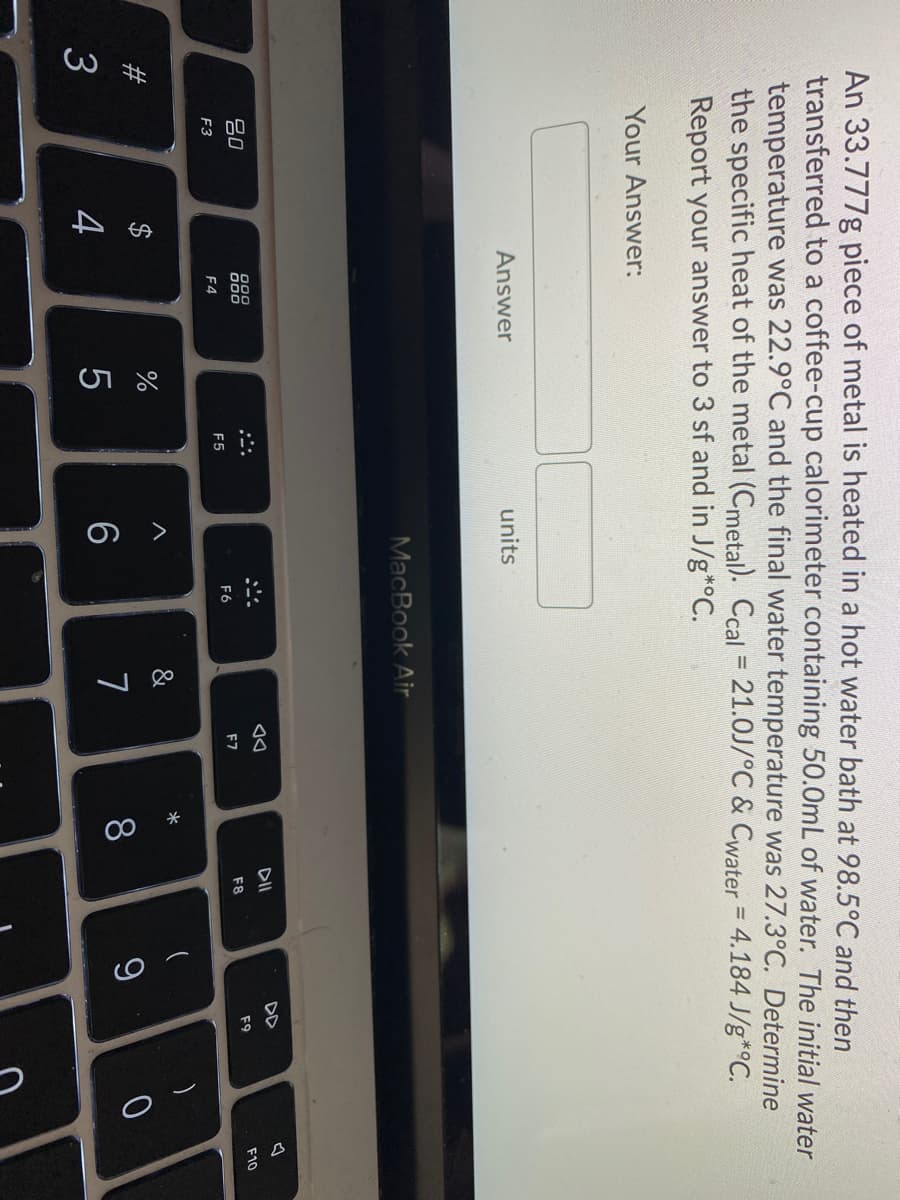
an 33.777g piece of metal is heated in a hot water bath at 98.5°C and then cransferred to a coffee-cup calorimeter containing 50.0mL of water. The initial water temperature was 22.9°C and the final water temperature was 27.3°C. Determine the specific heat of the metal (Cmetal). Ccal = 21.0J/°C & Cwater = 4.184 J/g*°C. %3D Report your answer to 3 sf and in J/g*°C. Your Answer:
an 33.777g piece of metal is heated in a hot water bath at 98.5°C and then cransferred to a coffee-cup calorimeter containing 50.0mL of water. The initial water temperature was 22.9°C and the final water temperature was 27.3°C. Determine the specific heat of the metal (Cmetal). Ccal = 21.0J/°C & Cwater = 4.184 J/g*°C. %3D Report your answer to 3 sf and in J/g*°C. Your Answer:
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter7: Chemical Energy
Section: Chapter Questions
Problem 109AE: A sample of nickel is heated to 99.8C and placed in a coffee-cup calorimeter containing 150.0 g...
Related questions
Question
Transcribed Image Text:%24
An 33.777g piece of metal is heated in a hot water bath at 98.5°C and then
transferred to a coffee-cup calorimeter containing 50.0mL of water. The initial water
temperature was 22.9°C and the final water temperature was 27.3°C. Determine
the specific heat of the metal (Cmetal). Ccal = 21.0J/°C & Cwater = 4.184 J/g*°C.
Report your answer to 3 sf and in J/g*°C.
Your Answer:
Answer
units
MacBook Air
DII
DD
000
吕0
F9
F10
F7
F8
F6
F5
F3
F4
*
#3
7
8
9
4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
