An Ag-Cu alloy contains 25% a and 75% B at 600°C. The composition of the overall alloy is approximately 1000 L+B 800 8-87 28-12 922 a +B 500 972 400 Ag 10 20 30 40 50 60 70 80 90 Cu %Cu (by weight) - A. 80.2% Cu В. 73.5% Cu C. 65.8% Cu D. 89.6% Cu
An Ag-Cu alloy contains 25% a and 75% B at 600°C. The composition of the overall alloy is approximately 1000 L+B 800 8-87 28-12 922 a +B 500 972 400 Ag 10 20 30 40 50 60 70 80 90 Cu %Cu (by weight) - A. 80.2% Cu В. 73.5% Cu C. 65.8% Cu D. 89.6% Cu
Precision Machining Technology (MindTap Course List)
2nd Edition
ISBN:9781285444543
Author:Peter J. Hoffman, Eric S. Hopewell, Brian Janes
Publisher:Peter J. Hoffman, Eric S. Hopewell, Brian Janes
Chapter2: Measurement, Materials, And Safety
Section2.7: Heat Treatment Of Metals
Problem 2RQ: Direct hardening can be performed on steel containing at least________ carbon.
Related questions
Question
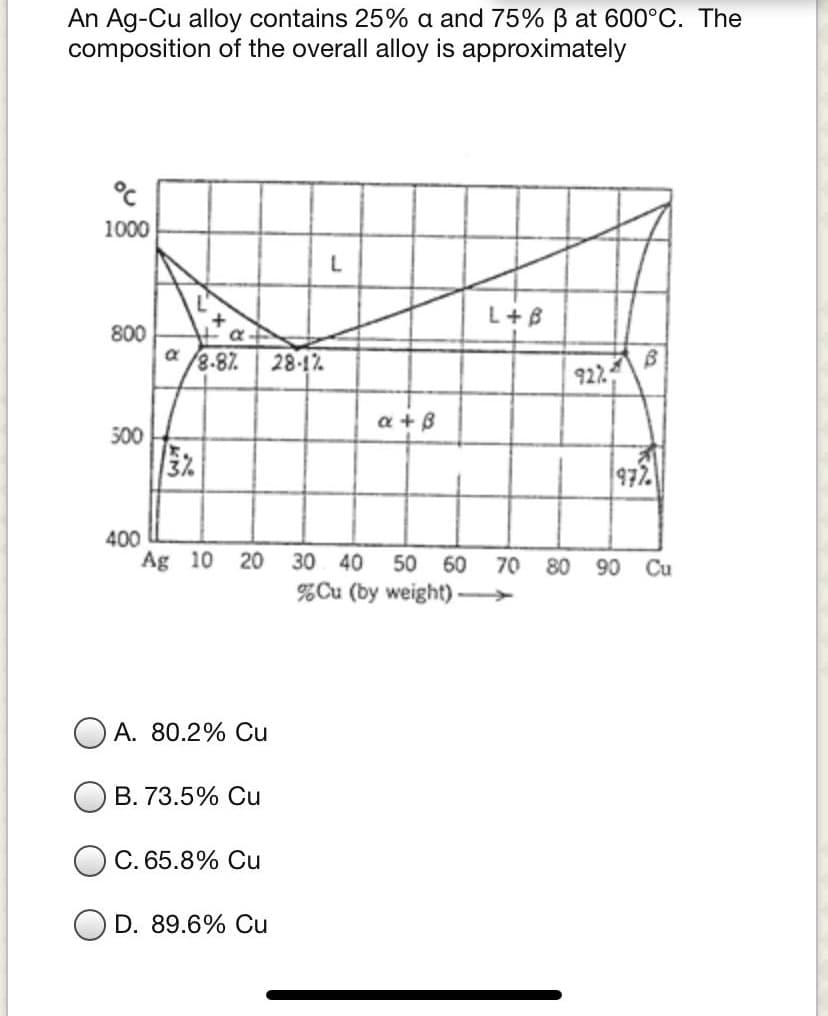
Transcribed Image Text:An Ag-Cu alloy contains 25% a and 75% B at 600°C. The
composition of the overall alloy is approximately
1000
L+B
800
8-87
28-12
922
a + B
500
972
400
Ag 10 20 30 40 50
60 70
80
90 Cu
%Cu (by weight)
A. 80.2% Cu
B. 73.5% Cu
C. 65.8% Cu
D. 89.6% Cu
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Precision Machining Technology (MindTap Course Li…
Mechanical Engineering
ISBN:
9781285444543
Author:
Peter J. Hoffman, Eric S. Hopewell, Brian Janes
Publisher:
Cengage Learning

Precision Machining Technology (MindTap Course Li…
Mechanical Engineering
ISBN:
9781285444543
Author:
Peter J. Hoffman, Eric S. Hopewell, Brian Janes
Publisher:
Cengage Learning