An enzyme has a single active site at which it can bind and hydrolyze either X or Y but the enzyme cannot bind X and Y at the same time. Which of the following statements are TRUE?
An enzyme has a single active site at which it can bind and hydrolyze either X or Y but the enzyme cannot bind X and Y at the same time. Which of the following statements are TRUE?
Biochemistry
9th Edition
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Chapter1: Biochemistry: An Evolving Science
Section: Chapter Questions
Problem 1P
Related questions
Question
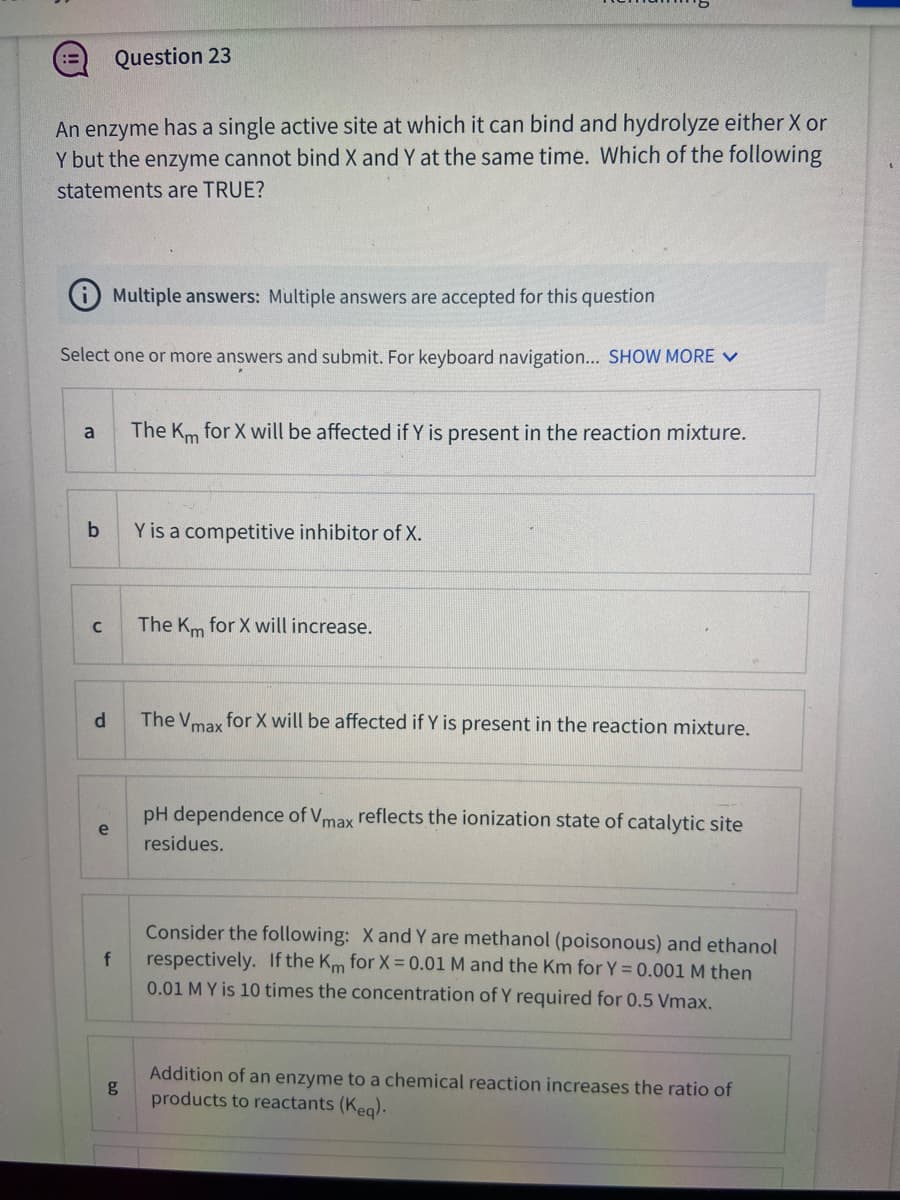
Transcribed Image Text:Question 23
An enzyme has a single active site at which it can bind and hydrolyze either X or
Y but the enzyme cannot bind X and Y at the same time. Which of the following
statements are TRUE?
Multiple answers: Multiple answers are accepted for this question
Select one or more answers and submit. For keyboard navigation... SHOW MORE V
The Km for X will be affected if Y is present in the reaction mixture.
a
Y is a competitive inhibitor of X.
The Km for X will increase.
d.
The V for X will be affected if Y is present in the reaction mixture.
max
pH dependence of Vmax reflects the ionization state of catalytic site
e
residues.
Consider the following: X and Y are methanol (poisonous) and ethanol
respectively. If the Km for X = 0.01 M and the Km for Y = 0.001 M then
0.01 M Y is 10 times the concentration of Y required for 0.5 Vmax.
Addition of an enzyme to a chemical reaction increases the ratio of
g
products to reactants (Keg).
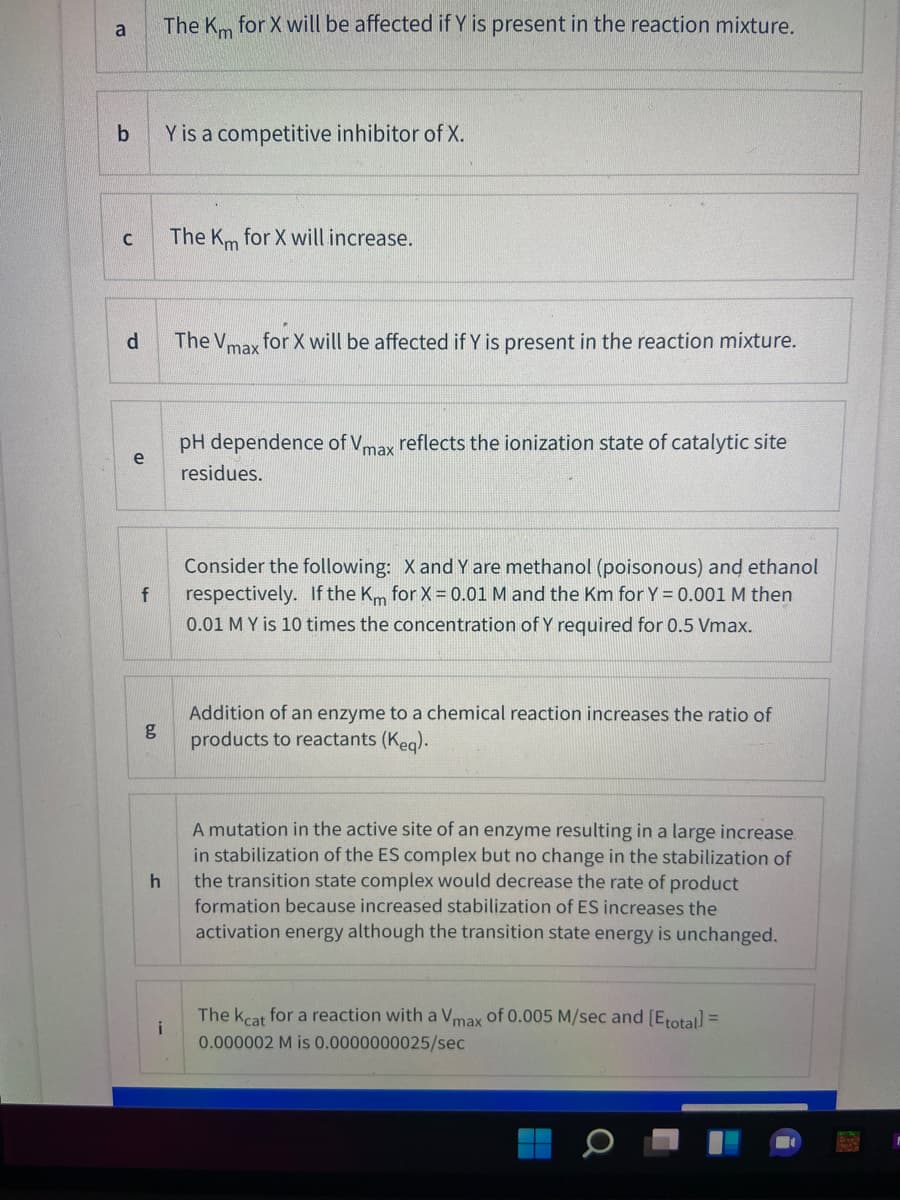
Transcribed Image Text:a
The Km for X will be affected if Y is present in the reaction mixture.
Y is a competitive inhibitor of X.
The Km for X will increase.
d.
The Vmax
for X will be affected if Y is present in the reaction mixture.
pH dependence of Vmax reflects the ionization state of catalytic site
e
residues.
Consider the following: X and Y are methanol (poisonous) and ethanol
respectively. If the Km for X = 0.01 M and the Km for Y = 0.001 M then
0.01 M Y is 10 times the concentration of Y required for 0.5 Vmax.
Addition of an enzyme to a chemical reaction increases the ratio of
g
products to reactants (Keg).
A mutation in the active site of an enzyme resulting in a large increase.
in stabilization of the ES complex but no change in the stabilization of
the transition state complex would decrease the rate of product
formation because increased stabilization of ES increases the
activation energy although the transition state energy is unchanged.
The kcat for a reaction with a Vmax of 0.005 M/sec and [Erotal =
i
0.000002 M is 0.0000000025/sec
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781319114671
Author:
Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:
W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:
9781464126116
Author:
David L. Nelson, Michael M. Cox
Publisher:
W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul…
Biochemistry
ISBN:
9781118918401
Author:
Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:
WILEY

Biochemistry
Biochemistry
ISBN:
9781319114671
Author:
Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:
W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:
9781464126116
Author:
David L. Nelson, Michael M. Cox
Publisher:
W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul…
Biochemistry
ISBN:
9781118918401
Author:
Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:
WILEY

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Fundamentals of General, Organic, and Biological …
Biochemistry
ISBN:
9780134015187
Author:
John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:
PEARSON