An Ochem student wanted to predict the major product of an alkene addition reaction using the molecules below in the presence of water and trace amount of strong acid. Choose the correct statement below. Molecule (i) forms a secondary carbocation and undergoes a methyl shift. Molecule (ii) forms a secondary carbocation and undergoes a hydride shift. Both molecules do not undergo carbocation rearrangement. Both molecules form secondary alcohols as major product
An Ochem student wanted to predict the major product of an alkene addition reaction using the molecules below in the presence of water and trace amount of strong acid. Choose the correct statement below. Molecule (i) forms a secondary carbocation and undergoes a methyl shift. Molecule (ii) forms a secondary carbocation and undergoes a hydride shift. Both molecules do not undergo carbocation rearrangement. Both molecules form secondary alcohols as major product
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter21: Chemistry Of The Main-group Elements
Section: Chapter Questions
Problem 21.2QP
Related questions
Question
An Ochem student wanted to predict the major product of an
|
Molecule (i) forms a secondary carbocation and undergoes a methyl shift. |
||
|
Molecule (ii) forms a secondary carbocation and undergoes a hydride shift. |
||
|
Both molecules do not undergo carbocation rearrangement. |
||
|
Both molecules form secondary alcohols as major product. |
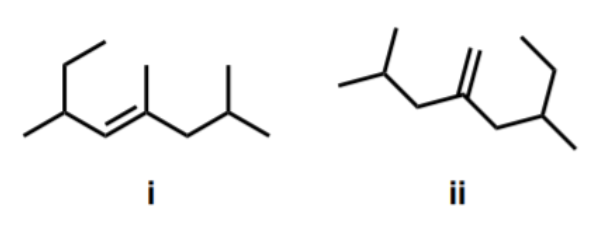
Transcribed Image Text:ii
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning
