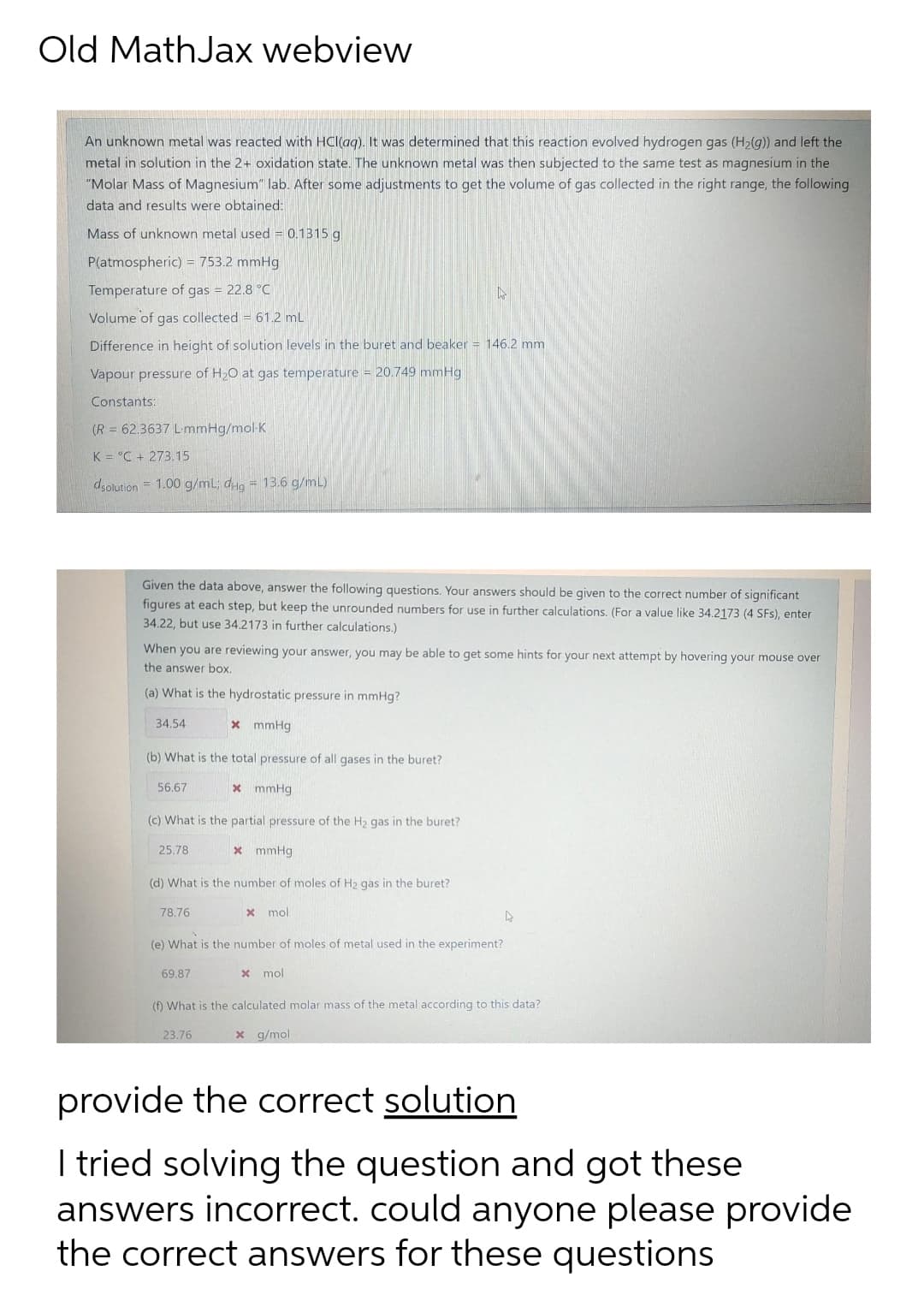
An unknown metal was reacted with HCl(ag). It was determined that this reaction evolved hydrogen gas (H2(g)) and left the metal in solution in the 2+ oxidation state. The unknown metal was then subjected to the same test as magnesium in the "Molar Mass of Magnesium" lab. After some adjustments to get the volume of gas collected in the right range, the following data and results were obtained: Mass of unknown metal used = 0.1315 g P(atmospheric) = 753.2 mmHg Temperature of gas = 22.8 °C Volume of gas collected = 61.2 mL %3D Difference in height of solution levels in the buret and beaker 146.2 mm Vapour pressure of H2O at gas temperature = 20.749 mmHg Constants: (R = 62.3637 L-mmHg/mol-K %3D K = °C + 273.15 dsolution = 1.00 g/mL; dHg = 13.6 g/mL)
An unknown metal was reacted with HCl(ag). It was determined that this reaction evolved hydrogen gas (H2(g)) and left the metal in solution in the 2+ oxidation state. The unknown metal was then subjected to the same test as magnesium in the "Molar Mass of Magnesium" lab. After some adjustments to get the volume of gas collected in the right range, the following data and results were obtained: Mass of unknown metal used = 0.1315 g P(atmospheric) = 753.2 mmHg Temperature of gas = 22.8 °C Volume of gas collected = 61.2 mL %3D Difference in height of solution levels in the buret and beaker 146.2 mm Vapour pressure of H2O at gas temperature = 20.749 mmHg Constants: (R = 62.3637 L-mmHg/mol-K %3D K = °C + 273.15 dsolution = 1.00 g/mL; dHg = 13.6 g/mL)
Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 5P
Related questions
Question
1
Transcribed Image Text:Old MathJax webview
An unknown metal was reacted with HCl(ag). It was determined that this reaction evolved hydrogen gas (H2(g)) and left the
metal in solution in the 2+ oxidation state. The unknown metal was then subjected to the same test as magnesium in the
"Molar Mass of Magnesium" lab. After some adjustments to get the volume of gas collected in the right range, the following
data and results were obtained:
Mass of unknown metal used = 0.1315 g
P(atmospheric) = 753.2 mmHg
Temperature of gas = 22.8 °C
Volume of gas collected = 61.2 mL
Difference in height of solution levels in the buret and beaker = 146.2 mm
Vapour pressure of H2O at gas temperature = 20.749 mmHg
Constants:
(R = 62,3637 LmmHg/mol-K
K = °C + 273.15
dsolution = 1.00 g/mL; dHg = 13.6 g/mL)
Given the data above, answer the following questions. Your answers should be given to the correct number of significant
figures at each step, but keep the unrounded numbers for use in further calculations. (For a value like 34.2173 (4 SFs), enter
34.22, but use 34.2173 in further calculations.)
When you are reviewing your answer, you may be able to get some hints for your next attempt by hovering your mouse over
the answer box.
(a) What is the hydrostatic pressure in mmHg?
34.54
x mmHg
(b) What is the total pressure of all gases in the buret?
56.67
* mmHg
(c) What is the partial pressure of the H2 gas in the buret?
25.78
* mmHg
(d) What is the number of moles of H2 gas in the buret?
78.76
x mol
(e) What is the number of moles of metal used in the experiment?
69.87
x mol
(f) What is the calculated molar mass of the metal according to this data?
23.76
x g/mol
provide the correct solution
I tried solving the question and got these
answers incorrect. could anyone please provide
the correct answers for these questions
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps

Recommended textbooks for you


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning



Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning