Analyze the following structure of farnesol in terms of isoprene units joined by head-to-tail linkages. wh TOH (1) How many isoprene units are there in a farnesol molecule? (2) Classify farnesol as a monoterpene (one terpene unit), a sesquiterpene (1.5 terpene units), or a diterpene (2 terpene units).
Analyze the following structure of farnesol in terms of isoprene units joined by head-to-tail linkages. wh TOH (1) How many isoprene units are there in a farnesol molecule? (2) Classify farnesol as a monoterpene (one terpene unit), a sesquiterpene (1.5 terpene units), or a diterpene (2 terpene units).
Chapter1: Lewis Structures
Section: Chapter Questions
Problem 26EQ: The skeleton is benzaldoxime is The number of valence electrons is as follows: from the phenyl...
Related questions
Question
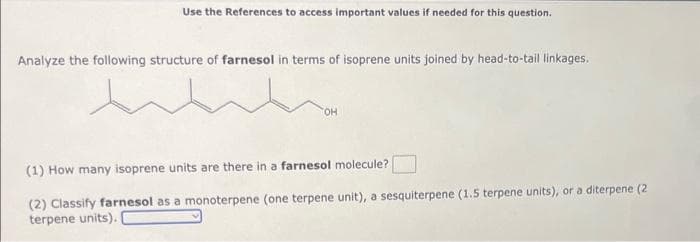
Transcribed Image Text:Use the References to access important values if needed for this question.
Analyze the following structure of farnesol in terms of isoprene units joined by head-to-tail linkages.
TOH
(1) How many isoprene units are there in a farnesol molecule?
(2) Classify farnesol as a monoterpene (one terpene unit), a sesquiterpene (1.5 terpene units), or a diterpene (2
terpene units).

Transcribed Image Text:Analyze the following structure of citral in terms of isoprene units joined by head-to-tail linkages.
w
Use the References to access important values if needed for this question.
(1) How many Isoprene units are there in a citral molecule?
(2) Classify citral as a monoterpene (one terpene unit), a sesquiterpene (1.5 terpene units), or a diterpene (2 terpene
units)
Subr
monoterpene
sesquiterpene
diterpene
Entire Group 9 more group attempts remaining
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning