Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D below each structure. ed A table of pK, values for various organic and inorganic acids can be found in the references section. CH disopropylamine disopropylamide butoxide a) The weaker base is b) Its conjugate acid is c) The species that predominate at equilibrium are (two letters, e.g. AC)
Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D below each structure. ed A table of pK, values for various organic and inorganic acids can be found in the references section. CH disopropylamine disopropylamide butoxide a) The weaker base is b) Its conjugate acid is c) The species that predominate at equilibrium are (two letters, e.g. AC)
Chapter9: Aqueous Solutions And Chemical Equilibria
Section: Chapter Questions
Problem 9.21QAP
Related questions
Question
8
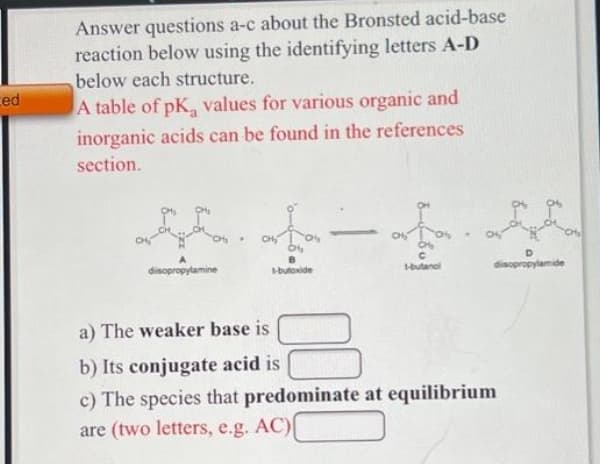
Transcribed Image Text:Answer questions a-c about the Bronsted acid-base
reaction below using the identifying letters A-D
below each structure.
ced
A table of pK, values for various organic and
inorganic acids can be found in the references
section.
CH
CH
B
disopropylamine
-butoxide
butanol
disopropylamide
a) The weaker base is
b) Its conjugate acid is
c) The species that predominate at equilibrium
are (two letters, e.g. AC)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you



Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning



Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT