Q: 3. Consider the electrophilic aromatic substitution reaction below. Draw three resonance structures…
A: More stabilized and Compound with every atom has complete octet shows most contribution towards…
Q: Draw molecules in their correct protonation state
A: Concept introduction: We know organic compounds can be seperated from the mixture simply by using…
Q: e a short note on how to perform the Acree-Rosenheim reaction test. Make sure to mention the…
A: The Acree-Rosenheim test is performed in the chemical laboratory. Generally, the active reagent for…
Q: For each statement below, label it as causing a faster or slower rate for an E1 reaction. If it…
A: E1 stands for unimolecular elimination reaction. Since, the rate of the reaction is proportional to…
Q: Please help me solve this synthesis. It takes 5 steps
A: OKAY, I will give complete synthesis by using 5 steps. You can see step by step solution. How to…
Q: но OH
A: The reaction given is,
Q: The powerful electron-withdrawing property of a carbonyl group increases the acidity of a-protons…
A: The electronegativity of the carbonyl group is highest among other functional groups.
Q: and II, propose a sequenced reaction, identifying the two (or more if you are bold) individual…
A: In Mannich reaction nucleophilic addition of an amine to a carbonyl group occurs. If the product…
Q: Draw the product of cyclohexanone, bromine and base catalysis. thank you for your explanation in…
A: The major product of the given reaction is illustrate here: Let the base is NaOH. We know that Br is…
Q: Rank the bold-faced hydrogens for the following compounds from most acidic to least acidic. Explain…
A: Lewis acids are defined as those acids which are able to lose a proton. Acidity is dependent on…
Q: Draw the organic product of the Bronsted acid-base reaction. Include all lone pairs and charges as…
A:
Q: scheme
A:
Q: Reaction of butylamine with hydrochloric acid
A: Given Reaction type = Neutralization reaction Reactant = Butylamine and HCl Product = to be…
Q: Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Supply the reagent for the…
A: The given reaction is: To identify the suitable reagent.
Q: エーロ Complete the curved arrow mechanism for this reaction (including the charges where necessary): H…
A: In an organic reaction mechanism, a curved arrow represents the transfer of electrons. In an organic…
Q: Provide the MAJOR product of the following reaction and draw the curved-arrow
A: Regiochemistry is the chemistry of regioselective reactions Regioselectivity is the priority of…
Q: So, in this lewis acid-base reaction, do we need to draw lone pairs or not? Why or why not? Thank…
A: In the given lewis acid base reaction : Is it necessary to represents the lone pair of electrons ?
Q: The product in this reaction is basic enough to be protonated by a dilute HCl solution. Draw the…
A:
Q: Arrange the intermediates below in order of increasing basicity:
A: The basicity is ability of species to accept proton. More the acceptance of proton, more is basic…
Q: Predict the product of the following substitution reaction?
A: Nucleophilic substitution biomolecular reaction (SN2): The bimolecular nucleophilic substitution…
Q: NaOH но. Н-О, Неat
A: When an acid and base react they form a salt and water. Here benzoic acid react with NaOH to form…
Q: Hello, can you write the explicit formulas of A, B, C and D in the synthesis below?
A:
Q: Draw the organic product of the following reaction.
A: In the given reaction, alkene is treated with Cl2 and the mechanism of the reaction is given in…
Q: Please clearly draw the overall reaction taking place between methyl salicylate and sodium…
A:
Q: Propose a reasonable multi-step synthetic route that would accomplish the following Ph
A:
Q: When compound A is treated with a strong base, a mixture of product B and C results. Which base…
A: Lithium base . It is two types 1. Less hindered Lithium base 2. Highly hindered Lithium base.
Q: Explain why alkylation of an α-carbon works best if the alkyl halide used in the reaction is a…
A: The alkylation reaction of an α-carbon depends on the R-X (alkyl halide) used in the reaction. The…
Q: Draw the appropriate fishhook arrows for the following reaction,
A:
Q: kindly answer a and b, asap. Thank you!
A: The structure of the given compound is :
Q: 1. BH3 THF 2. H₂O2/NaOH/H₂O ●
A:
Q: Provide mechanism with curved arrows for Part B: Synthesis of benzil
A: PhC(=O)-CH(IH)-Ph on oxidation with HNO3 gives Ph-C(=O)-C(=O)-Ph which is called benzil. At first…
Q: Circle the strongest acid in the following set of molecules. Provide a brief explanation (20 words…
A: "Since you have posted a question with multiple subparts, we will solve first three sub parts for…
Q: Complete the mechanism below with the missing structure and arrows. Also identify any acids, bases,…
A:
Q: In the picture, Each of the following compounds has more than one kind of (a-H's). First draw all…
A: see the picture uploaded in the solution. the most acidic proton is circled. if more than one…
Q: NH base Z-
A:
Q: Construct a three-step synthesis of 3-bromo-3-methyl-2-butanol from 2-methyl-2-butene by dragging…
A: Step-1 : 3-bromo-2-methylbutan-2-ol formation Step-2 : Epoxide formation Step-3 :…
Q: Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Which compound below undergoes…
A: Alkenes undergo addition reaction with HBr to form alkyl halide. The H+of HBr acts as an…
Q: The compound below exists in two tautomeric forms. Circle the most acidic proton in the keto form…
A: Keto and enol are form of tautomerism.
Q: а.) b.) с.)
A: Acetals on hydrolysis gives corresponding aldehyde or ketone and alcohols. Let's see here,
Q: Draw the major organic product of the Bronsted acid-base reaction. Include all lone pairs and…
A: A Bronsted acid donates a proton and a Bronsted base accepts a proton. Hence, in a Bronsted…
Q: Please provide a synthetic approach for the following reactions. Make sure to do the retrosynthetic…
A:
Q: Which compound in the following pair is more basic?
A: The resonance structures for pyrrole are given below - While resonance structures of piridine are…
Q: Complete this concept map to summarize all epoxide reactions: CH3 CH₂ НО. Weak Nuc, H₂O+ CH3 H3C 1.…
A:
Q: Provide a retrosynthetic analysis and forward synthesis for the compound shown below. and any other…
A:
Q: Two methods convert an alkyl halide to a carboxylic acid having onemore carbon atom. Depending on…
A: Two methods convert an alkyl halide to a carboxylic acid having one more carbon atom. Depending on…
Q: The SN2 reaction shows a reversible mechanism when the basic strength nucleophile and leaving group…
A: Generally SN2 reactions are substitution nucleophilic biomolecular reactions. The mechanism involves…
Q: In the window below, draw both the starting acid derivative and the ultimate carboxylic acid…
A: Benzoyl chloride when reacted with methane amine forms a tetrahedral alkoxide ion. The negative…
Q: Provide a plausible arrow pushing mechanism for the reaction below. Note that the starting material…
A: Applying basic rule of reagents and chemical reaction.
Q: Consider the below molecule and its hydrogen atoms labeled a through e. Rank the named hydrogen…
A: acidity - increase order - Hd<Hc<He<Ha<Hb
draw the major organic product of the Bronsted acid-base reaction. Include all lone pairs and charges as appropriate. Ignore any counterions.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

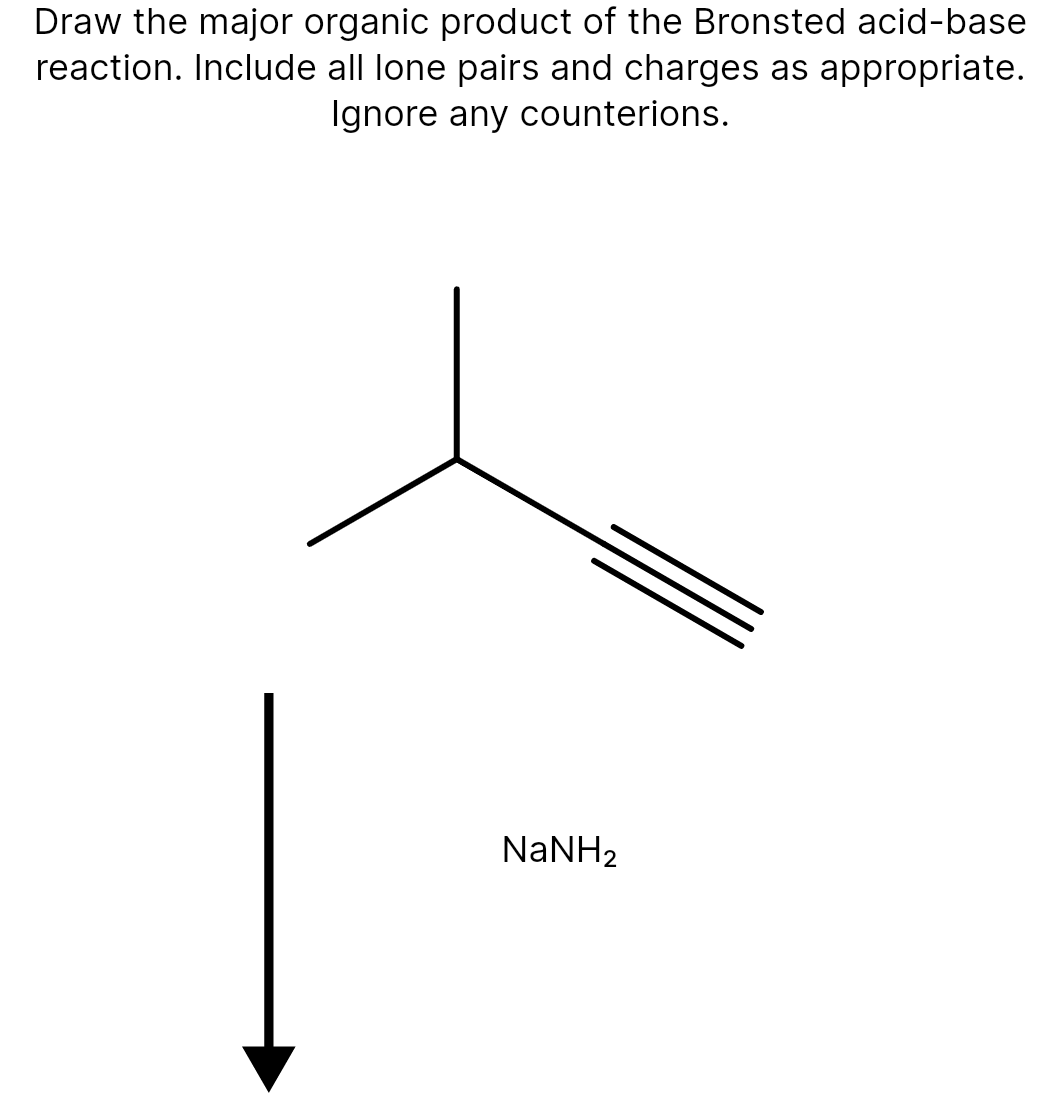
- K15. rank from weakest to strongest Bronsted base. Please give explanation and what the rankings look like for this questionDraw the major organic product of the Bronsted acid-base reaction. Include all lone pairs and charges. ----------NaNH2----------------->draw the major organic product of the Bronsted acid-base reaction. Include all lone pairs and charges as appropriate. Ignore any counterions.
- A) for each compound show its conjugate base. lone pairs have been left out. B) rank the conjugate base in the order you would predict, from most to least stable. C) rank the original compounds in order, from strongest to weakest acid.Propoxide (CH3CH2CH2O- ) is a larger molecule than ethoxide (CH3CH2O- ), yet they are equally basic. Explain why they are equally basic.1) For the following groups of molecules (labeled A-C), rank the basicity of the molecules in order from least basic (3) to most basic (1). Explain your reasoning.
- Which is a stronger base: RO- or RS-?Rank the following compounds in order of increasing acidity (1 = least acidic, 3 = most acidic) and in the space provided use resonance (of the conjugate base) to explain why the compound you have labelled “3” is the most acidic.Aside from H₃O⁺, the other product during the dissociation reaction of HN₃ is _____ and it is the _____ of HN₃. H₂N₃; acid H⁺; conjugate base N₃⁻; conjugate base N₃⁻; conjugate acid None of the given.
- Can someone please rank the hydrogen's on the third carbon from most acidic to least acidic?4. Circle the most basic nitrogen in the molecule and explain why it is more basic.For the following groups of molecules (labeled A-C), rank the acidity of the molecules in order from least acidic (3) to most acidic (1). Explain your reasoning.

