Arterial blood contains about 0.25 g of oxygen per liter at 37°C and standard atmospheric pressure. Under these conditions, the Henry's law constant is kH = 3.7 x 10-2 mol/(L • atm), and the mole fraction of O2 in the atmosphere is 0.209. 1st attempt Part 1 i See Periodic Table O See Hint Calculate the solubility (in M) of O2 in the blood of a climber on Mt. Everest, where Patm = 0.35 atm. Part 2 O See Hint Calculate the solubility (in M) of O2 in the blood of a scuba diver at a depth of 100 feet, where Patm = -3 atm. M
Arterial blood contains about 0.25 g of oxygen per liter at 37°C and standard atmospheric pressure. Under these conditions, the Henry's law constant is kH = 3.7 x 10-2 mol/(L • atm), and the mole fraction of O2 in the atmosphere is 0.209. 1st attempt Part 1 i See Periodic Table O See Hint Calculate the solubility (in M) of O2 in the blood of a climber on Mt. Everest, where Patm = 0.35 atm. Part 2 O See Hint Calculate the solubility (in M) of O2 in the blood of a scuba diver at a depth of 100 feet, where Patm = -3 atm. M
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter6: Solutions And Colloids
Section: Chapter Questions
Problem 6.29P
Related questions
Question
100%
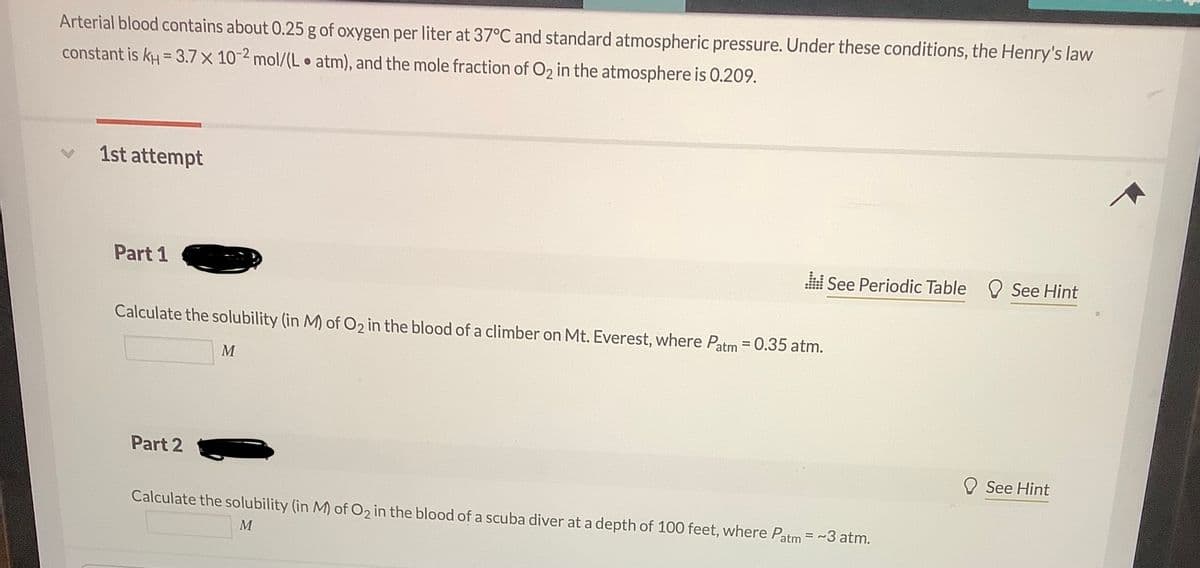
Transcribed Image Text:Arterial blood contains about 0.25 g of oxygen per liter at 37°C and standard atmospheric pressure. Under these conditions, the Henry's law
constant is kH = 3.7 x 10-2 mol/(L atm), and the mole fraction of O2 in the atmosphere is 0.209.
1st attempt
Part 1
See Periodic Table O See Hint
Calculate the solubility (in M) of O2 in the blood of a climber on Mt. Everest, where Patm = 0.35 atm.
%3D
M
Part 2
See Hint
Calculate the solubility (in M) of O2 in the blood of a scuba diver at a depth of 100 feet, where Patm = ~3 atm.
M
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning