At a festival, spherical balloons with a radius of 280. cm are to be inflated with hot air and released. The air at the festival will have a temperature of 25 C and must be heated to 100 °C to make the balloons float. 3.00 kg of butane (C,H10) fuel are available to be burned to heat the air. Calculate the maximum number of balloons that can be inflated with hot air. Here are some data you may find useful: Specific heat capacity of air: 1.009 8"C Density of air at 100 "C: 0.946 kg 1.898 Density of butane at 100 "C: m Formation enthalpy of butane at 25 °C:-125.7 mol Any other data you need should be taken from the ALEKS Data resource. balloons
At a festival, spherical balloons with a radius of 280. cm are to be inflated with hot air and released. The air at the festival will have a temperature of 25 C and must be heated to 100 °C to make the balloons float. 3.00 kg of butane (C,H10) fuel are available to be burned to heat the air. Calculate the maximum number of balloons that can be inflated with hot air. Here are some data you may find useful: Specific heat capacity of air: 1.009 8"C Density of air at 100 "C: 0.946 kg 1.898 Density of butane at 100 "C: m Formation enthalpy of butane at 25 °C:-125.7 mol Any other data you need should be taken from the ALEKS Data resource. balloons
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter12: Thermodynamic Processes And Thermochemistry
Section: Chapter Questions
Problem 3P
Related questions
Question
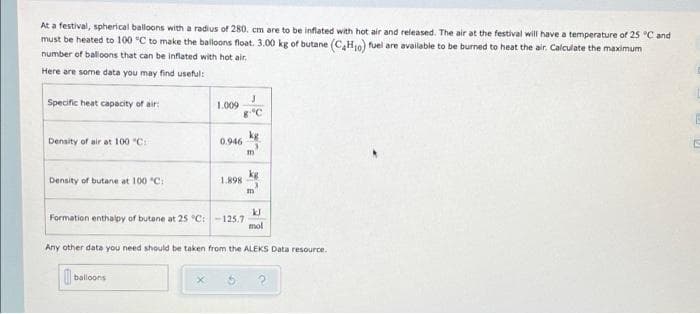
Transcribed Image Text:At a festival, spherical balloons with a radius of 280. cm are to be inflated with hot air and released. The air at the festival will have a temperature of 25 C and
must be heated to 100 °C to make the balloons float. 3.00 kg of butane (C,H10) fuel are available to be burned to heat the air. Calculate the maximum
number of balloons that can be inflated with hot air.
Here are some data you may find useful:
Specific heat capacity of air:
1.009
Density of air at 100 "C:
0.946
kg
1.898
Density of butane at 100 "C:
m
Formation enthalpy of butane at 25 °C:-125.7
mol
Any other data you need should be taken from the ALEKS Data resource.
| balloons
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning