At a molecular level, explain why in osmosis there is a net migration of solvent from the side of the membrane less concentrated in solute to the side more concentrated in solute by selecting all that apply. O The side of the membrane that is less concentrated in solute is more concentrated in solvent. O The side of the membrane that is less concentrated in solute is less concentrated in solvent. O The escaping tendency of the solvent increases with its concentration. O The difference in escaping tendency results in a net migration of solvent from the side less concentrated in solute to the side more concentrated in solute. O The difference in escaping tendency results in a net migration of solvent to the side less concentrated in solute from the side more concentrated in solute. O None of these. eTextbook and Media Attempts: 0 of 15 used Submit Answer Save for Later
At a molecular level, explain why in osmosis there is a net migration of solvent from the side of the membrane less concentrated in solute to the side more concentrated in solute by selecting all that apply. O The side of the membrane that is less concentrated in solute is more concentrated in solvent. O The side of the membrane that is less concentrated in solute is less concentrated in solvent. O The escaping tendency of the solvent increases with its concentration. O The difference in escaping tendency results in a net migration of solvent from the side less concentrated in solute to the side more concentrated in solute. O The difference in escaping tendency results in a net migration of solvent to the side less concentrated in solute from the side more concentrated in solute. O None of these. eTextbook and Media Attempts: 0 of 15 used Submit Answer Save for Later
Chapter11: Properties Of Solutions
Section: Chapter Questions
Problem 6RQ: In terms of Raoults law, distinguish between an ideal liquid-liquid solution and a nonideal...
Related questions
Question
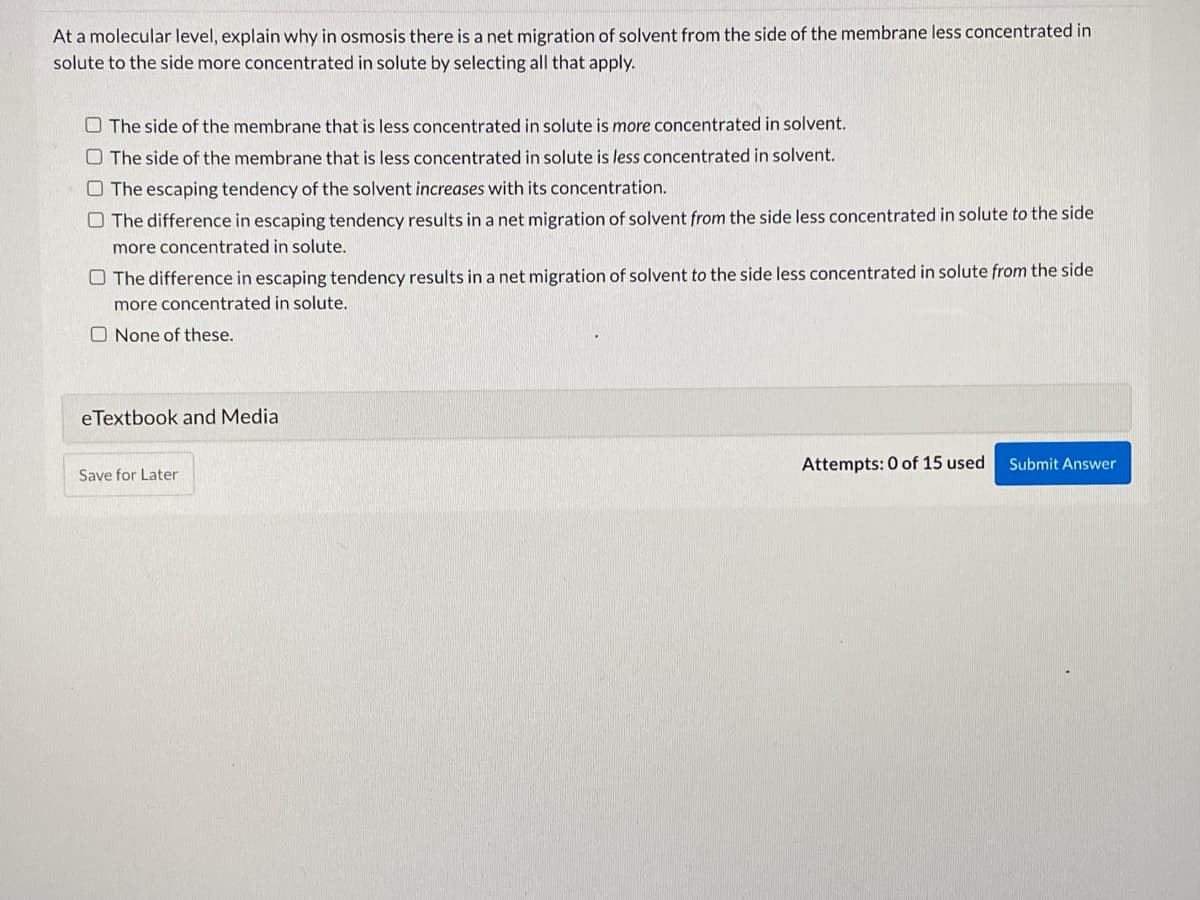
Transcribed Image Text:At a molecular level, explain why in osmosis there is a net migration of solvent from the side of the membrane less concentrated in
solute to the side more concentrated in solute by selecting all that apply.
O The side of the membrane that is less concentrated in solute is more concentrated in solvent.
O The side of the membrane that is less concentrated in solute is less concentrated in solvent.
O The escaping tendency of the solvent increases with its concentration.
O The difference in escaping tendency results in a net migration of solvent from the side less concentrated in solute to the side
more concentrated in solute.
O The difference in escaping tendency results in a net migration of solvent to the side less concentrated in solute from the side
more concentrated in solute.
O None of these.
eTextbook and Media
Attempts: 0 of 15 used
Submit Answer
Save for Later
Expert Solution
Step 1
Solution is made up of two components: solute and solvent. Substance which is present in less amount is called solute.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning