Q: 5: Using the following plot as a standard, how would Vmau Km, and the graphs look when.? Sobtrate…
A: Enzymes are biocatalyst which accelerate the biochemical reactions. Molecules on which enzymes act…
Q: TABLE 2: EFFECT OF TEMPERATURE ON CATALASE ACTIVITY STRENGTH OF REACTION Temperature…
A: The explanation is given below-
Q: The effect of reversible competitive inhibitor can be cancelled by: increasing the product…
A:
Q: Match the term with the defination: 1. The portion of the enzyme where catalysis occurs a. Enzyme b.…
A: the correct matching is as follows:
Q: Consider the following figure for the reaction A A+B C+D Progress of the reaction Is this reaction…
A: Catalysis is the process of altering the rate of a chemical reaction by adding a susbtance known as…
Q: Penicillin binds to transpeptidase thereby preventing enzyme activity. A) If penicillin binds at the…
A: Note : Since you have asked a question with multiple sub-parts, we will do the first three…
Q: What is the decomposition reaction of Hydrogen peroxide with catalase? OH2+CO2 + CATALASE -> H202 O…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: An enzyme whose KM is 10-4 M in the presence of a substrate concentration of 10-2 M It is capable of…
A:
Q: Which enzyme would catalyze the reaction illustrated below? triacylglycerol + 3H2O → glycerol + 3…
A: The reaction is triacylglycerol + 3H2O → glycerol + 3 fatty acids
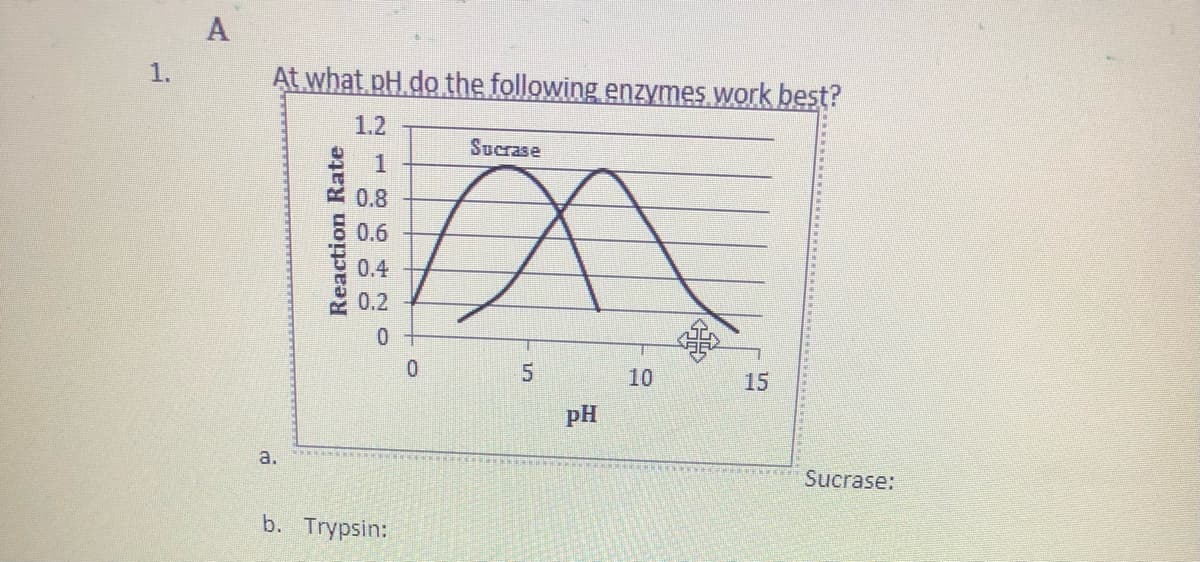
Q: With reference to enzyme action, explain what is happening in the diagram below based on the…
A: The activity of enzymes are based on pH as well as the on the reaction rate.
Q: Consider the following image depicting enzyme regulation for a two subunit enzyme. Ro Which of the…
A: Enzymes: The enzymes are known as the biological catalyst. Mostly, enzymes are made up of proteins.…
Q: One of the beolow statements is FALSE Amytase Pepain Arginese 4. 10 12 O a. Irreversible enzyme…
A: The given graph in the question is a plot of activity of an enzyme vs. pH of the medium. In this…
Q: From the given activity, 1.) What is the effect of temperature to the enzyme? 2.) What are some…
A: Hi, we are supposed to answer one question. To get the remaining questions solved please mention the…
Q: regarding a reversible E? fect can be decreased by increasing the substra embles the substrate's…
A: According to the question, we need to determine which of the statement regarding a reversible…
Q: Reaction 25.1 Sucrose (aq) + H2O (l) ⇄ Glucose (aq) + Fructose (aq) What is the Keq expression for…
A: When the rate of forward reaction is equal to the rate of backward reaction this state is called…
Q: CH,OH НО HO CH,OH CH2O ОН HO HOCH2 O НО OH НО ОН CH,OH II ОН I CH2O앵 НО HO HO CH,OH ОН CH2OH OH HO…
A:
Q: to cysteine residues in protein active sites. What glycolytic enzyme would be affected? As a result,…
A: A)In the range of metal concentrations analysed we found that only hexokinase and…
Q: 1. Draw a line graph to represent the data. Compare the two enzymes. % Activity of % Activity of…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: 23.Carbamyl phosphate synthetase_is a mitochondrial enzyme; it uses ammonium as the source of…
A: 23. correct option : A. 1, 2 Carbamyl phosphate synthetase 1 is a mitochondrial enzyme and uses…
Q: In the first step of aminoacyl group linkage to a tRNA, an amino acid reacts with ATP to yield an…
A: The difference between anhydride and mixed anhydride bonds is given in the table below:
Q: 8) what l;kely Caused in reaction the decline rate over time a) co-factor depletion b) substrate…
A: The rate of reaction depends on the change in concentration of the reactants over time and if the…
Q: If you wanted to decrease the deltaG of an enzyme-catalyzed reaction and could make only one change,…
A: Gibbs free energy is the free energy difference between the products and reactants of a chemical…
Q: 7. Which modification of the reaction environment will increase the rate of reaction in which an…
A: In a chemical reaction; the substance which involves in conversion is said to be reactant whereas…
Q: The activity of an enzyme requires a glutamic acid to display its –COOH functional group in the…
A:
Q: Q 19.6.3 In an experiment, the thermal degradation process of the enzyme produced the results in…
A: thermal degradation process of enzymes are first order reactions and for first order reaction: rate…
Q: 13. In the diagram of glycogen shown below, circle the substrates for glycogen debranching enzyme.…
A: The debranching enzyme is those substances that facilitate glycogen breakdown. The glycogen…
Q: 9. An example of an isoenzyme used for clinical diagnosis is a. lactate dehydrogenase b. creatine…
A: Lactate dehydrogenase isoenzymes (LDH) were used widely in the past for diagnosis of myocardial…
Q: 21.78 How will each of the following changes affect the rate of an enzyme-catalyzed reaction whose…
A: The activity of enzyme varies with temperature. The activity of enzyme increases to some extent with…
Q: As shown a proposed mechanism for carboxypeptidase A.(a) What is the role of Glu 270 in…
A: (a) Glutamine is present with carboxyl group which attract the hydroxyl linkage of the…
Q: Which of the following is the most reasonable explanation for why the curve on the graph below does…
A: Enzymes are biological catalysts. They can increase the rate of a reaction. The enzymes are mostly…
Q: Q3: Amylase in saliva begins the digestion of starch in the mouth . Would you expect this enzyme to…
A: A question based on general chemistry that is to be accomplished.
Q: 31) A compound that binds to the enzyme at a site other than the active site, and changes its shape…
A: (31) (A) Irreversible inhibitor - It inactive an enzyme by bonding covalently to a particular…
Q: 45. Which of the following statements about enzymes is true? A) Enzymes increase the activation…
A: 45. Correct Option : (D) Enzymes lower the transition state for the rate determining step.
Q: Chymotrypsin is secreted by the pancres and passed into the intestine. The optimal pH for this…
A: Chymotryspin is an enzyme used to break down the protein in the small intestine. Trypsin is used…
Q: Why do most living organisms have an enzyme to decompose hydrogen peroxide? Becâuse often hydrogen…
A: hydrogen peroxide forms as a product of metabolism
Q: An enzyme has an optimal pH of 7.1 and an optimal temperature at 37° C. Which statement describes a…
A: Given, An enzyme has an optimal pH of 7.1. An optimal temperature of 37oC. At a constant temperature…
Q: explain each graph above about enzymes and the factors affecting enzymatic activity.
A: Enzymes create chemical reactions in the body. They actually speed up the rate of a chemical…
Q: Enzyme activity can be controlled by availability of substrates and cofactors. U The binding of a…
A: Answer - The correct option is -(1) and (5) Solution - According to the question - proteolysis,…
Q: #6
A: The inhibition of urease in the presence of Hg, Cd or Cd (which forms metal ions) implies that ions…
Q: What is the important values needed to calculat Km and Kcat afor an enzyme data?
A: We have to tell the important values are needed to calculate the Km and Kcat for an enzyme catalyzed…
Q: Use the Michaelis-Menten equation to explain relationship between the amount of product produced and…
A:
Q: What impact on k1 and k-1 would be when a mutation in the enzyme (E) makes the interaction between E…
A: Here, if we consider the reaction between enzyme (E) and substrate (S), then : K 1 is the rate…
Q: Carbonic anhydrase is an enzyme that catalyzes the conversion of carbon dioxide to bicarbonate ion.…
A: Carbonic anhydrase catalyzes the conversion of carbon dioxide to bicarbonate ion. Carbonic anhydrase…
Q: QUESTION 2 List and describe the 4 different catalytic strategies that Serine proteases use to…
A: Serine proteases (or serine endopeptidases) are enzymes that cleave peptide bonds in proteins, in…
Q: Q 21: After the substrate diffuses with the enzyme, the next action in series is the A. amino acids…
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: 17.) In the left graph below, the dashed line (- - - -) represents what? В) Km A) Vmax C)…
A: Vmax is the maximum rate of reaction when enzyme reacts with substrate and Km is the Michael's…
Q: 5 and 6
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: The enzyme d-amino acid oxidase has avery high turnover number because the d-amino acids are…
A:
Q: II. Analysis. Given below is a schematic diagram for a simple analysis of a novel tetrasaccharide…
A: D- fructose B - glucose C- galactose A-ribose From TLC profile we can see that four…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- The enzyme d-amino acid oxidase has avery high turnover number because the d-amino acids are potentially toxic. The KM for the enzyme is in the range of 1 to2 mM for the aromatic amino acids and in the range of 15 to20 mM for such amino acids as serine, alanine, and the acidicamino acids. Which of these amino acids are the preferred substrates for the enzyme?. Relation between Reaction Velocity and Substrate Concentration: MichaelisMenten Equation: At what substrate concentration would an enzyme with a kcat of 30.0s−1 and a Km of 0.0050 M operate at one-quarter of its maximum rate?What is the important values needed to calculat Km and Kcat afor an enzyme data?
- By what factor is the rate of a reaction changed if an enzyme lowers the Ea by 2.0 kJ/mol at 37°C? 32.9 times 2.2 times 10.2 times 15.1 times 7.0 timesAn enzyme whose KM is 10-4 M in the presence of a substrate concentration of 10-2 M It is capable of transforming 20% of the substrate in 10 minutes. Calculate how much substrate is had transformed in 20 minutes.From the given activity, 1.) What is the effect of temperature to the enzyme? 2.) What are some generalizations and conclusions in this activity?
- How is the maximal rate of the enzyme changed in the presence of an inhibitor?The time that is required for an enzyme to convert one substrate molecule into one product molecule is _________ A. Km. B. 1/kcat. C. 1/Km. D. kcat. Which of the following does not apply to the concerted model for subunit behavior: A. Each subunit can exist in a relaxed (R) and taut (T) conformation. B. All subunits will be in either the R or the T conformation at the same time. C. Some subunits can be in the R state while others are in the T state. D. The presence of inhibitors will lead to more of the enzyme being in the T form E. The presence of activators will lead to more of the enzyme being in the R formHow can the Michaelis constant (Km) be identified and used to represent the affinity between the enzyme and the substrate?
- Two curves showing the rate versus substrate concentration are shown below for an enzymecatalyzed reaction. One curve is for the reaction in the presence of substance X. The other curve is for data in the absence of substance X. Examine the curves and tell which statement below is FALSE. A. X is an activator of the enzyme. B. The enzyme exhibits non-Michaelis-Menten kinetics. C. X is likely an allosteric effector. D. X is a competitive inhibitorA4 Your enzyme is inhibited by one of the compounds depicted above. The inhibitor for your specific protein is indicated via the PDB protein code shown.On the basis of the enzyme’s structure, its substrate and mechanism of action, predict what type of enzyme inhibition ( i.e., competitive or non-competitive) may occur and justify why you think this is the case . (Up to 50 words) TYPE NOT WRITE.answer from a to D a..Which of the enzyme has greater attraction for the subtrate b..Using the graph explain you enzyme in questionC..Which enzyme has greater maximum velocity (VMax)d .why doesnt the reaction rate continue to increase with subtrate concentration?



