b Answered:E Enthalpy - Chemistry x 101 Chem101 app.101edu.co Submit Question 21 of 29 Using the equations Si(s) + 2 Cl2 (g) → SICI4 (s) AH° = -640.1 kJ/mol 2 Fe (s) + 3 Cl2 (g) → 2 FeCl3 (s) AH° = -800.0 kJ/mol Determine the enthalpy for the reaction 3 SiCl: (s) + 4 Fe (s) → 4 FECI3 (s) + 3 Si (s). kJ/mol 1 3 4 8 9. x 100 +/- 8:49 PM O Type here to search 12/8/2019 LO
b Answered:E Enthalpy - Chemistry x 101 Chem101 app.101edu.co Submit Question 21 of 29 Using the equations Si(s) + 2 Cl2 (g) → SICI4 (s) AH° = -640.1 kJ/mol 2 Fe (s) + 3 Cl2 (g) → 2 FeCl3 (s) AH° = -800.0 kJ/mol Determine the enthalpy for the reaction 3 SiCl: (s) + 4 Fe (s) → 4 FECI3 (s) + 3 Si (s). kJ/mol 1 3 4 8 9. x 100 +/- 8:49 PM O Type here to search 12/8/2019 LO
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter2: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 2.79E: The enthalpy of combustion of diamond is -395.4 kJ/mol. C s, dia O2 g CO2 g Determine the fH of C...
Related questions
Question
Transcribed Image Text:b Answered:E Enthalpy - Chemistry x
101 Chem101
app.101edu.co
Submit
Question 21 of 29
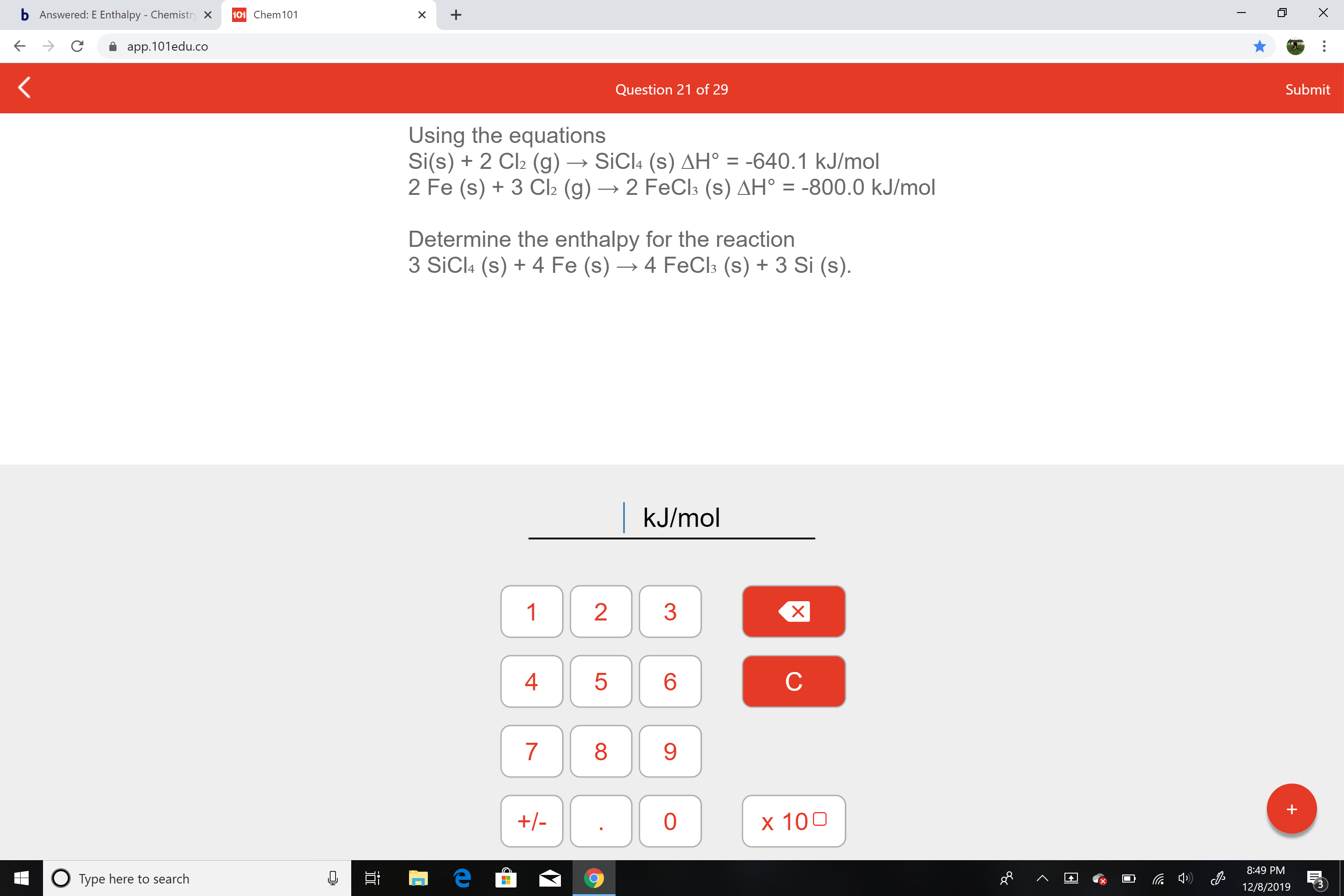
Using the equations
Si(s) + 2 Cl2 (g) → SICI4 (s) AH° = -640.1 kJ/mol
2 Fe (s) + 3 Cl2 (g) → 2 FeCl3 (s) AH° = -800.0 kJ/mol
Determine the enthalpy for the reaction
3 SiCl: (s) + 4 Fe (s) → 4 FECI3 (s) + 3 Si (s).
kJ/mol
1
3
4
8
9.
x 100
+/-
8:49 PM
O Type here to search
12/8/2019
LO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 4 images

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning