Q: By the action of a solution of hydrogen chloride in nitromethane, 3-methybut-1 - ene gives a mixture…
A: The reaction of 3-methylbut-1-ene and 3,3-dimethylbut-1-ene with a solution of hydrogen chloride in…
Q: For the reaction CO(g) + 3H2(g) → CH4(g) + H2O(g) AH = -206.1 kJ and AS° = -214.7 J/K The standard…
A: The objective of this question is to calculate the standard free energy change (ΔG°) for the given…
Q: None
A:
Q: One uses a 25.0 mL volumetric pipet to measure the volume of the buffer. The absolute error in this…
A: The objective of this question is to calculate the relative error in the measurement of the volume…
Q: 10) Use enthalpy of formation to calculate the heat of the following reactions. Write the…
A:
Q: An aqueous solution at 25 °C has a OH concentration of 0.002 M. Calculate the H₂O concentration. Be…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: 24. Water plays an important role in traditional ceramics shaping processes. With the help of a…
A: Step 1:The effect of water volume on the aggregate volume of clay-based ceramics is crucial in…
Q: Use the activities to calculate the molar solubility of Zn(OH)2 in 0.0167M K2SO4. What is the…
A: Step 1: Step 2: Step 3: Step 4:
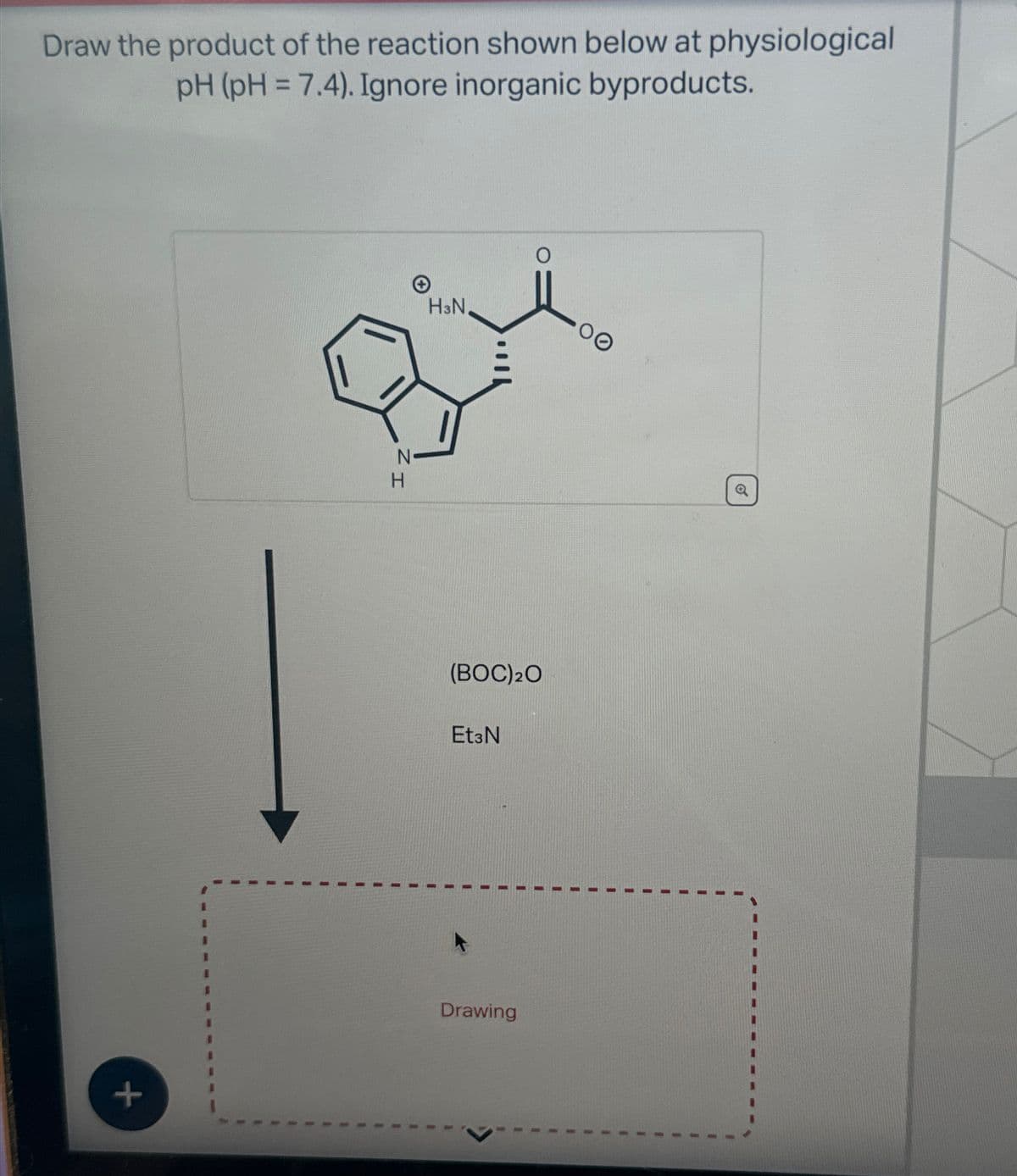
Q: Show the steps necessary to transform the acid on the left into the compound on the right. Be sure…
A: Answer:
Q: Predicting qualitatively how entropy changes with mixing and separation 1/3 For each system listed…
A: Step 1: Step 2: Step 3: Step 4:
Q: 4. Positron sources are used in PET scans (PET = positron emission tomography), which are an…
A: Step 1:4)C116→e01+B115Here a proton decays into a neutron by looking at the atomic number changes…
Q: Based on your ICE table (Part 2) and the definition of Ka, set up the expression for Ka in order to…
A: BCA TABLE:Step 1: Calculate the number of moles (mol) of HClO and NaClO by multiplying the…
Q: Calculate the volume in milliliters of a 0.75M barium acetate solution that contains 150 mmol of…
A: The objective of the question is to find the volume of a 0.75M barium acetate solution that contains…
Q: None
A: Thank you.
Q: Calculate the pH when 15.0 mL of 0.200 M HBr is mixed with 30.0 mL of 0.400 M CH₃NH₂ (Kb = 4.4 ×…
A: The objective of this question is to calculate the pH of a solution when a strong acid (HBr) is…
Q: A Suppose the molecule below is in acidic aqueous solution. Is keto-enol tautomerization possible? •…
A:
Q: Use Le Chatelier's principle to explain the change in pressure. Describe the difference in pressure…
A: Le Chatelier's principle portrays how a dynamic equilibrium alters to neutralize a change in…
Q: Calculate the volume in milliliters of a 1.8 mol / L; iron(3) bromide solution that contains 325.…
A: Step 1:We can calculate for the mL FeBr3 using dimensional analysis. First, I would like to convert…
Q: 6. Provide reasonable step-by-step mechanisms for the following reactions. Use arrows to indicate…
A:
Q: Please answer question below, be very deatiled and draw out the mechanism reaction with arrows
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw the major product of this reaction. Ignore inorganic byproducts.
A: Step 1: Step 2: Step 3: Step 4:
Q: The equilibrium constant, Kp, for the following reaction is 0.636 at 600 K. CoCl2(g) ? CO(g) +…
A: Step 1:Step 2:Step 3:
Q: Organic Chemistry Problem Please help. Thank you
A: SYNTHESIS:EXPLANATION:In the first step CN- attack the carbonyl carbon which is the electrophilic…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: pH i got from previous question which was calculate the pH of a 1.00L buffer solution that is 0.100M…
A: To calculate the pH of the buffer solution after the addition of NaOH, we need to consider the…
Q: NH3(g) + H2O(l) → NH2 + + H3O+ t The equilibrium constant is 1 x 10 -34. Is this reaction likely to…
A: The objective of the question is to determine whether the given reaction is likely to take place…
Q: Consider the reaction 2Fe(s) + 3Cl2(g) >>2FeCl3(s) Using standard thermodynamic data at 298K,…
A: Step 1: Given2Fe(s) + 3Cl2(g) → 2FeCl3(s)∆H°f (Fe) = 0 kJ/mol∆H°f (Cl2) = 0 kJ/mol∆H°f (FeCl3) =…
Q: None
A: The question is confusing as the proper reaction is not stated. If the reaction involves addition of…
Q: Use the data given in the table to calculate the value of AGxn at 25° C for the reaction described…
A: For #1: The Gibbs free energy change (ΔG0) of reaction can be calculated using the Gibbs free…
Q: Solid silver acetate is slowly added to 50.0 mL of a 0.0620 M sodium sulfite solution. The…
A: The objective of the question is to find the concentration of silver ion required to just initiate…
Q: Using examples, describe some typical levels of recent use for cocaine, benzoylecgonine and…
A: Recreational User (Vial Stored Frozen and Contains Preservatives):When a recreational user's…
Q: Strontium chloride and sodium fluoride react to form strontium fluoride and sodium chloride,…
A: The objective of this question is to find the volume of a 0.550 M NaF solution required to react…
Q: Of the following, which represents the intermediate that is most likely formed in the first step of…
A: The question is not clear whether the addition to the alkene is using Br2 or HBr. If it is…
Q: A Lewis base is also known as an electrophile. A)True B)False When water evaporates, the entropy of…
A: 1:A Lewis base is an electron donor. Lewis base has lone pair of electrons. Lewis base is known as…
Q: 127. Write a plausible reaction sequence of 4 steps starting from A, B, C, or D. You may use any…
A: The objective of the question is to design a plausible reaction sequence of 4 steps starting from…
Q: The IR provided is of monoacetylferrocene. Label each peak corresppnding to its appropriate…
A: In the IR spectrum of monoacetylferrocene, you'll typically observe several peaks corresponding to…
Q: Lead consists of four naturally occurring isotopes. Calculate the atomic mass for lead using the…
A: IsotopeMassAbundance (%)204.01.40206.024.10207.022.10208.052.40
Q: 1) Determine the Ka of a weak monoprotic acid. .00020 mole are dissolved in 100.0 mL solution. 20.0…
A:
Q: Consider an ideal gas enclosed in a 1.00 L container at an internal pressure of 24.0 atm. Calculate…
A: Step 1: first we have to find work done by the gas against external force.- pexternal(v2-v1). Step…
Q: Hybrid Orbitals Hybrid orbitals are formed by combining the valence orbitals on an atom. There are…
A: Step 1: There are 5 orbitals represented in this picture. Step 2: They are composed of 1s, 3p, 1d…
Q: dont provide handwriting solution...
A:
Q: Bromo cyclopentadiene reacts to form a Witting Reagent (A), but does not react with aldehydes or…
A: Here's the Structure A:
Q: None
A:
Q: For the reaction 2 H2(g) + O2(g) → 2 H₂O(g) which equation would be used to determine the entropy…
A: Step 1: Step 2: entropy change can be determined by calculating the change in number of gaseous…
Q: [References] Draw a structural formula for the organic product formed by treating butanal with the…
A: The product formed would be a silver complex with the butanal molecule. The silver ion would…
Q: How to calculate the pH of a buffer solution 1) Calculate the pH of a solution prepared by…
A: Step 1: Step 2: Step 3: Step 4:
Q: Multiple Attempts Not allowed. This test can only be taken once. Force Completion This test can be…
A:
Q: Zinc reacts with hydrochloric acid according to the reaction equation Zn(s) + 2HCl(aq) → ZnCl2(aq) +…
A: The objective of this question is to determine the volume of 2.50 M HCl(aq) required to react…
Q: onsider the reactionN2(g) + 2O2(g)2NO2(g)Using the standard thermodynamic data in the tables linked…
A: Step 1:Consider the given reaction N2(g)+2O2(g)---->2NO2(g) .The standard free energy change of…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- One of the heptanedione isomers below has a pka of approximately 9 rather than 20. Which isomer? Why? Draw its conjugate base and offer a full explanation of its acidity.Not sure how to approach this problem.Give only typing answer with explanation and conclusion What are the schemes for the following reactions?: Schemes: 1.Benzoic acid in NaOH 2.Acetic acid in NaHCO3 3.Propan-2-amine in water
- Show the two step dissociation process for H2SO4. Do both acidic hydrogens dissociate 100%?Base from the illustration: 1. Which compound/s will test positive in Baeyer’s test? 2. Which compound/s will produce an orange precipitate upon reaction with 2,4-DNPH? 3. Which compound/s will test positive in the Iodoform test? 4. Which compound/s will produce a brick-red precipitate upon reaction with Fehling’s reagent (CuSO4, tartrate, NaOH)? 5.Acid Halide Preparation reaction mechanism: Please use HCl and SOCl2 as reagents
- complete this reaction Fe³= ........agent (from-to) S2O3= .........agrnt (from-to).10.2 Complete the following reactions: Show the step-by-step process. Do not use shortcut methods. Make it as detailed as it can be. Encode (not hand-written)!In each of the following reactions, one of the reagents contains two reactive groups (either acidic or basic). You have to decide which group will be affected under mentioned conditions. To answer this problem correctly I suggest that you use the following strategy: (i) Write the products of the reaction: conjugate acid and conjugate base. Use curved arrows to track the flow of electrons; (ii) Using Table 3.1 of the textbook estimate and write pKa values for all acidic groups you have in the reaction equation including conjugate acid; (iii) If you wrote the correct reaction the estimated pKa values should confirm that the reaction transformation changes stronger acid to weaker acid and stronger base to weaker.
- Show the product formed as a result of the reaction between propanoic acid and benzylalcohol in an acidic environment by writing the reaction mechanism together.how to make derivative of 1) oxalic acid 2) benzophenone give reaction and name of derivative , please ASAPIn step 2 of the reaction sequence HCl is added to the basic reaction mixture after the reflux period. Show a reaction (with full structures) that explains the purpose of the HCl.

