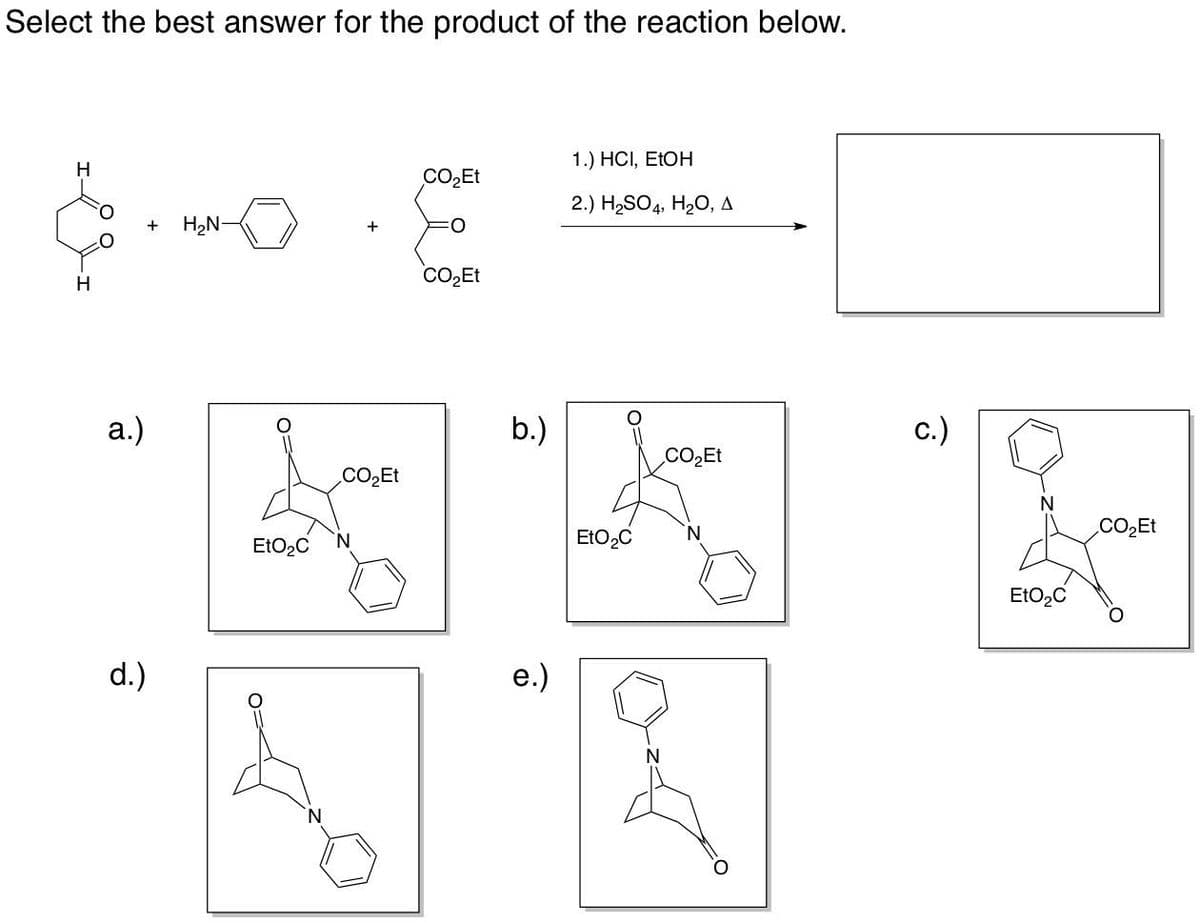
Select the best answer for the product of the reaction below. H a.) d.) 1.) HCI, EtOH COཉྫEt 2.) H,SO4, H¢O, A + HཉྫN- COཉྫEt EtOęC N N COཉྫEt b.) e.) EtO,C c.) CO,Et 0 EtOzC COཉྫEt
Q: Decide whether each of the molecules in the table below is stable, in the exact form in which it is…
A: Step 1: Step 2: Step 3: Step 4:
Q: Ph + ? Ph. B:0 Ph 0 Ph Ph Ph What is ? • Draw Mechanism
A:
Q: One uses a 25.0 mL volumetric pipet to measure the volume of the buffer. The absolute error in this…
A: The objective of this question is to calculate the relative error in the measurement of the volume…
Q: I need help with part b
A: 2. After adding 15 mL of KOH: At this point, 15 mL of 0.100 M KOH reacts with the barbituric acid.…
Q: The structure of the amino acid tyrosine is shown below in its completely protonated form. PK 16 OH…
A: The question is asking about the structure of the amino acid tyrosine in its completely protonated…
Q: Draw the Lewis structure and describe the bonding in Cr2(CH3COO)4(H2O)2 using MO theory. In the…
A: Step 1: To start, let's draw the Lewis structure for Cr2(CH3COO)4(H2O)2. Since we're using…
Q: Suppose 2.06 g of lead(II) acetate is dissolved in 250. mL of a 48.0 m M aqueous solution of…
A: Step 1:
Q: 20. Draw the line-angle formulas for the products from the hydrolysis of each of the following: a. +…
A: Step 1: The given reaction follows base-catalyzed hydrolysis of amides. In base-catalyzed hydrolysis…
Q: A 131.7 mg sample of powdered Carbamazepine tablets was boiled with ethanol, filtered and then made…
A: Step :
Q: While cleaning, you accidentally add water to a flask with non-halogenated solvent. Where should you…
A: Non-halogenated solvents, such as acetone or ethyl acetate, are organic compounds. When you…
Q: Attempts Force Completion This test can be saved and resumed later. Your answers are saved…
A: The question is asking us to determine the direction of the shift in equilibrium when a base (NaOH)…
Q: Calculate the activity coefficients for Ca+2 and SO42- in a solution containing 0.003 M CaSO4. You…
A: Step :
Q: Question 17 Which one of the following molecules has only 120° bond angles? tricky A BF3 B) SF6 C)…
A: Step 1: Step 2: Step 3: Step 4:
Q: Consider a U-shaped tube containing two aqueous solutions of ions, represented by orange spheres,…
A: The question is asking us to determine if the system is in equilibrium and to predict the direction…
Q: Cengage Learning OWLv2 | Online teaching and C…
A: The objective of this question is to calculate the entropy change for the system when 2.46 moles of…
Q: how much powder should I add to 250 mL of juice knowing that 226.798 grams of powder should be…
A: The objective of the question is to find out how much powder should be added to 250 mL of juice…
Q: If a 32 g bar of chocolate was completely combusted (to CO2 and H2O) with a stoichiometric…
A: Combustion of chocolate in presence of oxygen will show reaction like this C45H66O9 + O2 -> CO2 +…
Q: Assume that each compound in the drug mixture would make a spot on the paper the same size as those…
A: The objective of the question is to determine if the resolution of the paper chromatography method…
Q: (7 points) Calculate the silver ion concentration, [Ag*], of a solution prepared by dissolving 8.25…
A: Step 1:To find the silver ion concentration [Ag+],in the solution after dissolving 8.15g of AgNO3…
Q: 4. Determine the appropriate reagents to achieve the following transformations. 요 Η Ph Ph Η
A: Step 1:To achieve the transformations shown in the image, we need to determine the appropriate…
Q: The equilibrium constant, Kp, for the following reaction is 0.636 at 600 K. CoCl2(g) ? CO(g) +…
A: Step 1:Step 2:Step 3:
Q: An analytical chemist is titrating 187.2 mL of a 0.7600M solution of butanoic acid (HC3H7CO2) with a…
A: To calculate the pH of the solution during the titration, we can follow these steps:1. Calculate the…
Q: 3) Venlafaxine is a Wyeth antidepressant that is synthesized on a commercial scale from the starting…
A: Step 1:
Q: None
A: In CS2, the molecule consists of a central carbon atom bonded to two sulfur atoms by double bonds.…
Q: Predicting qualitatively how entropy changes with mixing and separation For each system listed in…
A: 1. Seawater:Change: Separation into freshwater and brine using a reverse osmosis filter.ΔS: ΔS >…
Q: Draw bond line structures for the following (a.) 4-Isopropyldecane (b.) 2,2-dichloropropane (c.)…
A:
Q: Cengage Learning OWLv2 | Online teaching and b Cengage Learning OWLv2 | O ☑ Funding Application…
A:
Q: Please don't provide handwritten solution ....
A: GivenMassM1 =0.125 kg (unknown)M2=0.285 kg (water)M3=0.150kg(calorimeter)Temparature,T1=…
Q: Cengage Learning OWLv2 | Online teaching and b Cengage Learning OWLv2 | O ☑ Inbox (4,532) -…
A: The objective of the question is to calculate the equilibrium constant for the given reaction at a…
Q: (11) Draw the major organic product of the reaction. Follow this procedure: determine whether the…
A: Step 1: Step 2: Step 3: Step 4:
Q: Complete the following reactions by drawing the major product(s) for each of the following…
A: The objective of the question is to predict the major product of the given chemical reaction. The…
Q: A buffer solution contains 0.373 M NaH2PO4 and 0.228 M K2HPO4. Determine the pH change when 0.048…
A: To determine the pH change when 0.048 mol HBr is added to 1.00L of a buffer solution containing…
Q: dont provide handwriting solution...
A: do leave a thumbs-up, as it motivates us to assist you better and faster. Thank You.
Q: Assume that each compound in the drug mixture would make a spot on the paper the same size as those…
A: Step 1: No, it's unlikely that paper chromatography would be sufficient to separate all the…
Q: P
A:
Q: 17.39 Using ethanol as the source of all the carbon atoms, describe efficient syntheses of each of…
A: Step :
Q: Predicting qualitatively how entropy changes with mixing and separation 1/3 elld For each system…
A: Do not forget to leave a helpful rating.Thank you and happy learning on Course Hero!
Q: Which of the following determines the direction of spontaneity? A Go Only option (B) Both (A) and…
A:
Q: Bromo cyclopentadiene reacts to form a Witting Reagent (A), but does not react with aldehydes or…
A: Here's the Structure A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts.
A: Step 1: Explanation Step 2: The answer is:Major product:
Q: 19.73 Propose an efficient synthesis for each of the following transformations: (f) Br H
A: Step 1: To synthesize 3-methylpentanal from 2-bromo-3-methylpentane, you can follow the steps…
Q: 8. (20 pt) Draw a mechanism for step 1 and a product & mech with a 2nd equiv. of MeMgBr. Me-MgBr…
A: The objective of the question is to understand the reaction mechanism involving Methylmagnesium…
Q: Please color the answer
A: If you further need clarification, comment please Thanks
Q: 2. Consider the following reaction: 4 HCN(/) + 5 O2(g) → 2 H₂O(g) + 4 CO₂(g) + 2 N2(g) (a) Using the…
A:
Q: 3- During the Aldol condensation experiment you add 1.7 mL of benzaldehyde (MW - 106.12 g/mol,…
A:
Q: Of the following, which represents the intermediate that is most likely formed in the first step of…
A: The question is not clear whether the addition to the alkene is using Br2 or HBr. If it is…
Q: Explain the similarities and differences between physical and chemical reactions with regards to…
A: The objective of this question is to understand the similarities and differences between physical…
Q: None
A: a) This molecule has two functional groups, the C=O i.e. ketone and the methyl CH3 group.b) This…
Q: b write a mechanism for the reaction using curved arrows to show electron reorganization.…
A: Step 1: Step 2: Step 3: Step 4:
Q: 2. The following table gives the values of the preferential adsorption (Qe) and respective…
A: Ce (mol/L)Qe (mol/g-charcoal)0.01814.660 x 10-40.88272.479 x 10-3The concentration of the…
Step by step
Solved in 2 steps with 1 images

- 2 A. could there be secondary product? B. Indicate why mud crystal is used in the reaction.1.how can i solve this question. 2.How is this type of problem named? How can I search for information on the Internet? 3.I know that the top of the arrow is the reagent and the bottom is the solvent. Can you tell me the process of finding them? Is it a problem that can be solved because I memorized reagents or solvents in advance?Where does the 40% and 60% product come from? How to determine this?

